Advertisements
Advertisements
प्रश्न
When a mixture of gases X and Y is compressed to 300 atm pressure and then passed over a catalyst consisting of a mixture of zinc oxide and chromium oxide (heated to a temperature of 300°C), then an organic compounds Z having the molecular formula CH4O is formed. X is a highly poisonous gas which is formed in appreciable amounts when a fuel burns in a limited supply of air; Y is a gas which can be made by the action of a dilute acid on an active metal; and Z is a liquid organic compound which can react with sodium metal to produce hydrogen gas.
(a) What are X, Y and Z?
(b) Write a balanced chemical equation of the reaction which takes place when X and Y combine to form Z. Indicate the conditions under which the reaction occurs.
Advertisements
उत्तर
(a) X is carbon monoxide gas (CO); it is formed when a fuel burns in limited supply of air. Y is hydrogen gas (H2); it is formed by the action of dilute acid on active metal. Z is methanol (CH3OH); it is a liquid organic compound that can react with sodium metal to produce hydrogen gas.
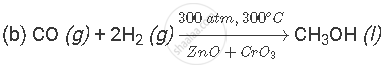
APPEARS IN
संबंधित प्रश्न
Translate the following statement into chemical equation and then balance it.
Hydrogen gas combines with nitrogen to form ammonia
Translate the following statement into chemical equation and then balance the equation:
Phosphorus burns in oxygen to give phosphorus pentoxide.
Balance the given chemical equation:
NH3 → N2 + H2
What do you understand by exothermic reactions?
An acid which can decolourise purple coloured potassium permanganate solution is:
(a) sulphuric acid
(b) citric acid
(c) carbonic acid
(d) hydrochloric acid
A colourless lead salt, when heated, produces a yellow residue and brown fumes.
(a) Name the lead salt.
(b) Name the brown fumes.
(c) Write a chemical equation of the reaction involved.
Write word equation for the following skeletal equation:
\[\ce{Zn + HCl -> ZnCl2 + H2}\]
Balance the following equation Also name the product formed.
`"K" + "CI"_2 → "KCI"`
Balance the following equation:
H2O + Cl2 → HCl + O2
Balance the following equation:
P + HNO3 → NO2 + H2O + H3PO4
Balance the following equation:
Zn + HNO3 `→` Zn(NO3)2 + H2O + NO2
Write the balanced chemical equation of the following reaction.
potassium dichromate + hydrochloric acid → Potassium chloride + chromium chloride + water + chlorine
Identify the type of reaction taking place of the following case and write the balanced chemical equation of the reaction:
Zinc reacts with silver nitrate to produce zinc nitrate and silver.
What is thermit process?
Give word equation for the following chemical reaction and give the names of the product formed.
\[\ce{CuO + H2SO4 -> CuSO4 + H2O}\]
Write word equation for the following chemical reaction given below. Also state the observation seen in the case.
Pb(NO3)2 + 2NH4OH → 2NH4N03 + Pb(OH)2↓
Give an example of a chemical equation in which two reactants form:
four product
Balance the following simple equation:
KHCO3 → K2CO3 + H2O + CO2
Balance the following simple equation:
Fe2O3 + CO → Fe + CO2
Underline the compound in the equation given below, it is incorrectly balanced and write the correct balancing for the same.
NaOH + CO2 → Na2CO3 + H2O
