Advertisements
Advertisements
प्रश्न
When a mixture of gases X and Y is compressed to 300 atm pressure and then passed over a catalyst consisting of a mixture of zinc oxide and chromium oxide (heated to a temperature of 300°C), then an organic compounds Z having the molecular formula CH4O is formed. X is a highly poisonous gas which is formed in appreciable amounts when a fuel burns in a limited supply of air; Y is a gas which can be made by the action of a dilute acid on an active metal; and Z is a liquid organic compound which can react with sodium metal to produce hydrogen gas.
(a) What are X, Y and Z?
(b) Write a balanced chemical equation of the reaction which takes place when X and Y combine to form Z. Indicate the conditions under which the reaction occurs.
Advertisements
उत्तर
(a) X is carbon monoxide gas (CO); it is formed when a fuel burns in limited supply of air. Y is hydrogen gas (H2); it is formed by the action of dilute acid on active metal. Z is methanol (CH3OH); it is a liquid organic compound that can react with sodium metal to produce hydrogen gas.
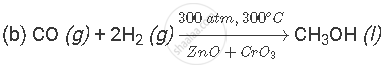
APPEARS IN
संबंधित प्रश्न
Write complete balanced equation for the following reaction:
Sulphur dioxide (gas) + Oxgyen (gas)  Sulphur trioxide (gas)
Sulphur trioxide (gas)
What are the various ways in which a chemical equation can be made more informative? Give examples to illustrate your answer.
Write any two observations in an activity which may suggest that a chemical reaction has taken place. Give an example in support of your answer.
Write the balanced equations for the following reaction, and add the state symbols:
Magnesium carbonate reacts with hydrochloric acid to produce magnesium chloride, carbon dioxide and water.
State one characteristic of the chemical reaction which takes place when wax is burned in the form of a candle?
The chemical equations are balanced to satisfy one of the following laws in chemical reactions. This law is know as:
(a) law of conservation of momentum
(b) law of conservation of mass
(c) law of conservation of motion
(d) law of conservation of magnetism
Write word equation for the following skeletal equation:
\[\ce{CO + O2 -> CO2}\]
Write your observation for the following chemical reaction and name the product formed:
When sugar is heated.
Write symbolic representation for the following word equation and balance them :
Iron + Sulphur → Iron sulphide.
Balance the following equation. Also name the products formed.
`"Mg" + "O"_2 → "MgO"`
Balance the following equation:
MnO2 + HCl → MnCl2 + H2O + Cl2
Balance the following equation:
C + H2SO4 → CO2 + H2O + SO2
Write word equation for the following molecular equation:
\[\ce{CuSO4 + 2NaOH -> Na2SO4 + Cu(OH)2↓}\]
Word equation:
State the colour of the products.
In certain reaction a change of state is observed i.e. solid to liquid, liquid to gas etc. – State the change of state of the products – to give the respective reactant.
\[\ce{2H2O ->[electric][current] 2H2 + O2}\]
Balance the following simple equation:
H2 + Cl2 → HCl
Balance the following simple equation:
H2S + Cl2 → S + HCl
Give a balanced equation by partial equation method, [steps are given below].
The reaction of excess ammonia with chlorine – Ammonia as a reducing agent
- Ammonia first reacts with chlorine to give hydrogen chloride and nitrogen.
- Hydrogen chloride then further reacts with excess ammonia to give ammonium chloride.
Write scientific reason.
Adding zinc particles to a solution of copper sulphate makes the blue solution colorless.
When aqueous solutions of potassium iodide and lead nitrate are mixed an insoluble substance separates out. The chemical equation for the reaction involved is:
