Advertisements
Advertisements
Question
What happens to the mass number of an element when (i) an α -particle, (ii) a β -particle, and (iii) γ -radiation is emitted?
Advertisements
Solution
- When an α particle is emitted mass number decreases by 4.
- When a β particle is emitted there is no change in the mass number.
- When a γ radiation is emitted there is no change in the mass number.
APPEARS IN
RELATED QUESTIONS
Name the radiations which are absorbed by greenhouse gases in the earth’s atmosphere.
What are free electrons?
Arrange α, β, and γ rays in ascending order with respect to their
1) Penetrating power.
2) Ionising power
3) Biological effect
State two similarities and two dissimilarities between the γ-rays and X-rays.
In following fig. shows a mixed source S of alpha and beta particles in a thick lead container. The particles pass through a magnetic field in a direction perpendicular to the plane of paper (inwards as shown by x). State and show in the diagram how the particles get affected.
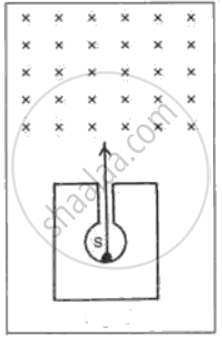
During the emission of a beta particle, the ______ number remains same.
Why should a radioactive substance not be touched by hands?
Name the three main parts of a Cathode Ray Tube.
Which of the following statements is/are correct?
- α particles are photons
- Penetrating power of γ radiation is very low
- Ionization power is maximum for α rays
- Penetrating power of γ radiation is very high
The mean lives of a radioactive substance are 1620 and 405 years for β-emission and β-emission respectively. The time after which three fourth of a sample will decay if it is decaying both by β-emission and β-emission simultaneously will be ______ years.
(Take ln 2 = 0.693)
