Advertisements
Advertisements
Question
What do you understand by the term radioactivity?
Advertisements
Solution
The process of self, spontaneous and random emission of a or P and y radiations from the nucleus of elements of atomic number higher than 82, is called radioactivity.
RELATED QUESTIONS
When does the nucleus of an atom tend to become radioactive?
Under which conditions does the nucleus of an atom tend to radioactive?
What kind of change takes place in a nucleus when a β -particle is emitted?
In following fig. shows a mixed source S of alpha and beta particles in a thick lead container. The particles pass through a magnetic field in a direction perpendicular to the plane of paper (inwards as shown by x). State and show in the diagram how the particles get affected.
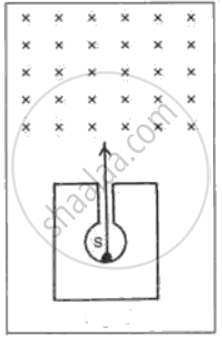
Justify with reason, whether the following nuclear reactions are allowed or not.
\[\ce{^A_Z X -> ^A_{Z + 1}Y + ^0_{-1}β}\]
State three common properties of beta rays and cathode rays.
State one use of radio-isotopes.
The half-life of a radioactive nuclide is 100 hours. The fraction of original activity that will remain after 150 hours would be ______.
In U238 ore containing Uranium the ratio of U234 to Pb206 nuclei is 3. Assuming that all the lead present in the ore is final stable product of U238. Half life of U238 to be 4.5 × 109 years and the age of ore is ______ × 109 years. (in 109 years)
A radioctive sample decays `7/8` times its original quantity in 15 minutes. The half-life of the sample is ______.
