Advertisements
Advertisements
प्रश्न
What do you understand by the term radioactivity?
Advertisements
उत्तर
The process of self, spontaneous and random emission of a or P and y radiations from the nucleus of elements of atomic number higher than 82, is called radioactivity.
संबंधित प्रश्न
Answer the following questions based on a hot cathode ray tube.
What will happen to the beam when it passes through the electric field?
What are free electrons?
What is meant by radioactivity?
In the following fig. shows a radioactive source S in a thick lead container. The radiations pass through an electric field between the plates P and Q. Complete the diagram to show the paths of α , β and γ radiations.
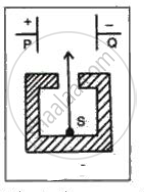
Why is the source S placed in a thick lead container?
A radioactive nucleus `""_"Z"^"A"` X first emits a beta particle and then an alpha particle to give the resulting nucleus `""_"Q"^"P"` Y What will be the values of P and Q in terms of A and Z?
A nucleus is \[\ce{^24_11 Na}\] β-radioactive.
Write the equation represent β-decay.
State three ways of obtaining an electron-beam.
Mention one important function of ear main parts:
State the principle on which the functioning of a nuclear reactor is based.
The half-life of a radioactive nuclide is 100 hours. The fraction of original activity that will remain after 150 hours would be ______.
