Advertisements
Advertisements
प्रश्न
Advertisements
उत्तर
Artificial radioactive substances can be produced by bombarding lighter nuclides with alpha particles, protons and neutron. The radioactive substances produces in this manner are called radioisotopes.
Radioisotopes can be used as:
(i) Rays from Radium produce satisfactorily improvement in skin diseases.
(ii) Radioactive Sulphur S35 helps to study advantages and disadvantages of fungicides.
APPEARS IN
संबंधित प्रश्न
Answer the following questions based on a hot cathode ray tube.
What will happen to the beam when it passes through the electric field?
How can they be made to leave the metal surface? (State any two ways)
In a cathode ray tube, what is the function of anode?
Write one use of cathode ray tube.
Under which conditions does the nucleus of an atom tend to radioactive?
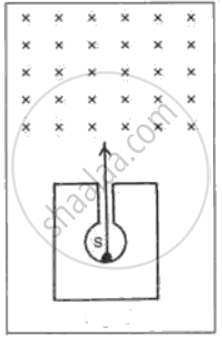
In following fig. shows a mixed source S of alpha and beta particles in a thick lead container. The particles pass through a magnetic field in a direction perpendicular to the plane of paper (inwards as shown by x). State and show in the diagram how the particles get affected.
What do you mean by electron emission?
Which of the following statements is/are correct?
- α particles are photons
- Penetrating power of γ radiation is very low
- Ionization power is maximum for α rays
- Penetrating power of γ radiation is very high
A radioactive nucleus \[\ce{^A_ZX}\] undergoes spontaneous decay in the sequence \[\ce{^A_ZX -> _{Z-1 }B -> _{Z-3}C -> _{Z-2}D }\], where Z is the atomic number of element X. The possible decay particles in the sequence are ______.
A radioactive element is placed in an evacuated chamber. Then the rate of radioactive decay will ______.
