Advertisements
Advertisements
प्रश्न
In a cathode ray tube, what is the function of anode?
Advertisements
उत्तर
In a cathode ray tube, the anode not only accelerates the electrons but also focuses them in a fine energetic beam.
APPEARS IN
संबंधित प्रश्न
A radioactive substance is oxidized. Will there be any change in the nature its radioactivity? Give a reason for your answer
Represent the change in the nucleus of a radioactive element when a β particle is emitted.
A nucleus `""_11^24Na` emits a beta particle to change into Magnesium (Mg)
(i) Write the symbolic equation for the process.
(ii) What are numbers 24 and 11 called?
(iii) What is the general name of `""_12^24Mg `with respect to `""_11^24Na` ?
What do you mean by Atomic number
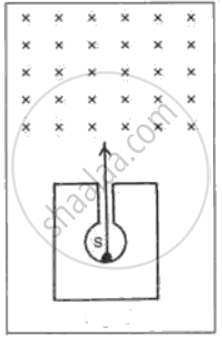
In following fig. shows a mixed source S of alpha and beta particles in a thick lead container. The particles pass through a magnetic field in a direction perpendicular to the plane of paper (inwards as shown by x). State and show in the diagram how the particles get affected.
A nucleus is \[\ce{^24_11 Na}\] β-radioactive.
Write the equation represent β-decay.
In a cathode ray tube state the purpose of covering cathode by thorium and carbon.
88Ra226 experiences three α - decay. Find the number of neutrons in the daughter element.
Two radioactive sources A and B of half lives of 1 hour and 2 hours, respectively, initially contain the same number of radioactive atoms. At the end of two hours, their rates of disintegration are in the ratio of ______.
A radioactive element is placed in an evacuated chamber. Then the rate of radioactive decay will ______.
