Advertisements
Advertisements
Question
The rate constant for a first order reaction is 60 s−1. How much time will it take to reduce the initial concentration of the reactant to its `1/16`th value?
Advertisements
Solution
t = `2.303/k log_10 ([A]_0)/([A])`
= `2.303/60 log_10 ([A]_0)/(1/16 xx [A]_0)`
= 0.3838 log10 (16)
= 0.3838 × 1.2041
= 4.62 × 10−2 s
APPEARS IN
RELATED QUESTIONS
Sucrose decomposes in acid solution into glucose and fructose according to the first order rate law with `t_(1/2)` = 3 hours. What fraction of the sample of sucrose remains after 8 hours?
For the decomposition of azoisopropane to hexane and nitrogen at 543 K, the following data are obtained.
| t (sec) | P(mm of Hg) |
| 0 | 35.0 |
| 360 | 54.0 |
| 720 | 63.0 |
Calculate the rate constant.
The time required for 10% completion of a first order reaction at 298 K is equal to that required for its 25% completion at 308 K. If the value of A is 4 × 1010 s−1. Calculate k at 318 K and Ea.
Following data are obtained for reaction :
N2O5 → 2NO2 + 1/2O2
| t/s | 0 | 300 | 600 |
| [N2O5]/mol L–1 | 1.6 × 10-2 | 0.8 × 10–2 | 0.4 × 10–2 |
1) Show that it follows first order reaction.
2) Calculate the half-life.
(Given log 2 = 0.3010, log 4 = 0.6021)
Define order of reaction. How does order of a reaction differ from molecularity for a complex reaction?
Which of the following graphs is correct for a first order reaction?




With the help of an example explain what is meant by pseudo first order reaction.
First order reaction is 50% complete in 1.26 × 1014s. How much time could it take for 100% completion?
A first order reaction is 50% complete in 20 minute What is rate constant?
In a first order reaction the concentration of reactants decreases from 400mol L-1 to 25 mol L-1 in 200 seconds. The rate constant for the reaction is ______.
Observe the graph shown in figure and answer the following questions:
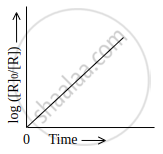
- What is the order of the reaction?
- What is the slope of the curve?
- Write the relationship between k and t1/2 (half life period).
A definite volume of H2O2 undergoing spontaneous decomposition required 22.8 c.c. of standard permanganate solution for titration. After 10 and 20 minutes respectively the volumes of permanganate required were 13.8 and 8.25 c.c. The time required for the decomposition to be half completed is ______ min.
The slope in the plot of ln[R] vs. time for a first order reaction is ______.
The slope in the plot of `log ["R"]_0/(["R"])` Vs. time for a first-order reaction is ______.
Write the unit of rate constant [k] for the first order reaction.
