Advertisements
Advertisements
Question
The best way to recover sugar from an aqueous sugar solution is :
Options
evaporation to dryness
distillation
filtration
crystallisation
Advertisements
Solution
crystallisation
Crystallisation is a better technique to recover sugar from sugar solution because sugar decomposes or gets charred on heating. Also, the soluble impurities get removed by crystallisation.
APPEARS IN
RELATED QUESTIONS
Gases have definite volume but no definite shape.
What are the characteristics of matter?
During respiration, glucose and oxygen enter our body cells and waste products carbon dioxide and water leave the body cells by the process of :
Name a metal which is soft and a non-metal which is hard.
Name one solid, one liquid and one gaseous non-metal.
What are the two types of pure substances? Give one example of each type.
How would you confirm that a colourless liquid given to your is pure water ?
What is the difference between solutions and colloids ?
You have been given a suspension and a solution. How could you tell the difference between them by their appearance ?
One of the following does not show Tyndall effect. This one is :
Name the process by which the various gases of the air are separated.
Which of the following mixture cannot be separated by using water as the solvent ?
The dyes present in fountain pen ink can be separated by the technique of :
Justify your answer.
| Figure 1 | Figure 2 | Figure 3 |
 |
 |
 |
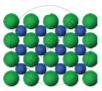
The arrangement of particles in three different phases of matter is shown above.
- Which state is represented by Fig. 1?
- In which state will the inter-particle attraction be maximum?
- Which one of them cannot be contained in an open vessel?
- Which one can take the shape of its container?
Celsius scale is a scale of temperature in which zero represents the melting point of ice and 100 represents the boiling point of water.
Complete the following table:
| S.No. | CELSIUS | KELVIN |
| 1. | 90°C | 363 K |
| 2. | ______ | 283 K |
| 3. | 63 °C | ______ |
| 4. | 250°C | ______ |
| 5. | ______ | 303 K |
The movement of particles in liquids is ______.
Gas-solid is ______.
Define the term matter.
Define the term matter.
