Advertisements
Advertisements
Question
State two physical properties on the basis of which metals can be distinguished from non-metals.
Advertisements
Solution
| Properties | Metals | Non-metals |
| Lustre | Lustrous | Non-lustrous |
| Heat and electrical conductivity | Good conductor | Poor conductor |
APPEARS IN
RELATED QUESTIONS
Define the term matter. What is it composed of?
Name the terms for the change of a solid into liquid.
What are molecules ?
Explain why air is used to inflate tyres.
Classify the following as elements or compounds :
Iron, Iron sulphide, Sulphur, Chalk, Washing soda, Sodium, Carbon, Urea
What are the three groups into which all the elements can be divided ? Name two elements belonging to each group.
One of the following substances is neither a good conductor of electricity nor an insulator. This substance is :
What happens when the temperature of a saturated sugar solution is increased ?
State whether the following statement is true or false :
Sponge is an example of solid sol.
Explain what happens when a beam of light is passed through a colloidal solution.
What is a suspension ? Give two examples of suspensions.
The solubility of ammonium chloride in water at various temperatures is given below :
| Temperature | : | 10°C | 20°C | 40°C | 60°C | 80°C |
| Solubility | : | 24 g | 37 g | 41 g | 55 g | 66 g |
What mass of ammonium chloride would be needed to make a saturated solution of ammonium chloride in fifty grams of water at 40°C ?
Describe the method of separating a mixture containing common salt, sand and ammonium chloride.
How is water purified on a large scale at water works ? Explain with the help of a labelled diagram. Name the substance which is added to kill germs in the drinking water supply ?
You are given a mixture of iodine in alcohol called tincture iodine. Which method will you use to recover both, iodine as well as alcohol, from this mixture ?
Justify your answer.
| Figure 1 | Figure 2 | Figure 3 |
 |
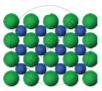 |
 |
The arrangement of particles in three different phases of matter is shown above.
- Which state is represented by Fig. 1?
- In which state will the inter-particle attraction be maximum?
- Which one of them cannot be contained in an open vessel?
- Which one can take the shape of its container?
Complete the following table:
| S.No. | CELSIUS | KELVIN |
| 1. | 90°C | 363 K |
| 2. | ______ | 283 K |
| 3. | 63 °C | ______ |
| 4. | 250°C | ______ |
| 5. | ______ | 303 K |
The movement of particles in liquids is ______.
Solids do not flow.
Define the term matter.
