Advertisements
Advertisements
Question
State how electrons are distributed in an atom. Explain in brief the rules which govern their distribution.
Advertisements
Solution
(a) Electrons revolve around the nucleus in imaginary paths called shells or orbits. Shells start from the nucleus to outwards.
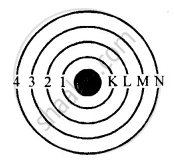
Rules: The maximum number of electrons in a shell is given by 2n2. Where n is the number of shell i.e. 1st shell can have a maximum of 2 electrons.
2n2 = 2(1)2 = 2 × 1 = 2
2nd shell can have maximum of 8 electrons
2n2 = 2(2)2 = 2 × 4 = 8
3rd shell can have maximum of 18 electrons
2n2 = 2 (3)2 = 2 × 9 = 18 and so on….
(b) Outer most orbit cannot have more than 8 electrons and 18 in penultimate orbit.
(c) A new shell cannot start until previous is filled completely.
APPEARS IN
RELATED QUESTIONS
Give the following a suitable word/phrase.
Protons and neutrons present in the nucleus.
What are the two main parts of which an atom is made of?
Tick the most appropriate answer.
Which of the following scientists observed that cathode rays consists of negatively charged particles?
- John Dalton
- J J Thomson
- James Chadwick
- Democritus
WRITE SHORT ANSWER
What is an atom?
WRITE SHORT ANSWER
Describe a discharge tube.
WRITE SHORT ANSWER
What do you mean by electron transition?
Name the sub-atomic particle whose charge is 0.
With reference to the discovery of the structure of an atom, explain in brief – William Crookes experiment for the discovery of cathode rays, followed by – J.J. Thomsons experiment pertaining to the constituents of the cathode rays. State which sub-atomic particle was discovered from his experiment.
Explain in brief – Goldstein’s experiment which led to the discovery of the proton.
Draw a neat labelled diagram representing an atom. Name the three sub-atomic particles in the atom and represent them symbolically showing the mass and charge of each. State where the sub-atomic particles are present in the atom.
