Advertisements
Advertisements
प्रश्न
State how electrons are distributed in an atom. Explain in brief the rules which govern their distribution.
Advertisements
उत्तर
(a) Electrons revolve around the nucleus in imaginary paths called shells or orbits. Shells start from the nucleus to outwards.
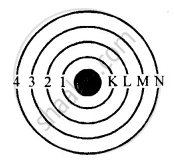
Rules: The maximum number of electrons in a shell is given by 2n2. Where n is the number of shell i.e. 1st shell can have a maximum of 2 electrons.
2n2 = 2(1)2 = 2 × 1 = 2
2nd shell can have maximum of 8 electrons
2n2 = 2(2)2 = 2 × 4 = 8
3rd shell can have maximum of 18 electrons
2n2 = 2 (3)2 = 2 × 9 = 18 and so on….
(b) Outer most orbit cannot have more than 8 electrons and 18 in penultimate orbit.
(c) A new shell cannot start until previous is filled completely.
APPEARS IN
संबंधित प्रश्न
Write true or false for the following statement
The central pad of the atom is called nucleus.
What are the two main parts of which an atom is made of?
FILL IN THE BLANK
The electrons revolving in shells close to the nucleus are called ................... electrons.
FILL IN THE BLANK
In an atom, the last shell containing electrons is called its..................shell.
FILL IN THE BLANK
The γ-rays are............................ radiations, like light.
Tick the most appropriate answer.
Which of the following scientists observed that cathode rays consists of negatively charged particles?
- John Dalton
- J J Thomson
- James Chadwick
- Democritus
Tick the most appropriate answer
β-particles are actually a stream of
- protons
- electrons
- neutrons
- none of these
Name the sub-atomic particle whose charge is 0.
Name the three fundamental particles of an atom.
Select the correct answer from the choice in the bracket to complete the sentence:
An element ‘X’ has six electrons in its outer or valence shell. Its valency is __________.
