Advertisements
Advertisements
Question
State following four properties each of α, β and γ radiations:
- Nature,
- Charge,
- Mass and
- Effect of electric field.
Advertisements
Solution
| Property | α - particle | β - particle | γ - particle |
| Nature | Stream of positively charged particles, i.e. helium nuclei | Stream of negatively charged particles, i.e. energetic electrons. | Highly energetic electromagnetic radiation. |
| Charge | Positive charge (Two times that of a proton) = + 3.2 x 10-19 C (or +2e) | Negative charge = - 1.6 x 10-19 C (or -e) | No charge |
| Mass | Four times the mass of proton i.e., 6.68 x 10-27 kg | Equal to the mass of electron, i.e. 9.1 x 10-31 kg | No mass (Rest mass is zero) |
| Effect of electric field | Less deflected | More deflected than alpha particles but in direction opposite to those of α particles | Unaffected |
APPEARS IN
RELATED QUESTIONS
The diagram in figure shows a radioactive source S placed in a thick lead walled container. The radiations given out are allowed to pass through a magnetic field. The magnetic field (shown as ×) acts perpendicular to the plane of paper inwards. Arrows shows the paths of the radiation A, B and C.
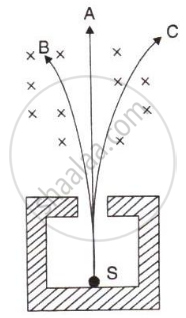
- Name the radiations labelled A, B and C.
- Explain clearly how you used the diagram to arrive at the answer in part (a).
In following Figure shows a radioactive source S in a thick lead walled container having a narrow opening. The radiations pass through an electric field between the plates A and B.
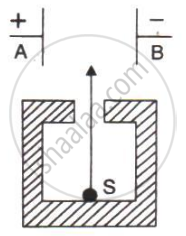
- Complete the diagram to show the paths of α, β and γ radiations.
- Why is the source S kept in a thick lead walled container with a narrow opening?
- Name the radiation which is unaffected by the electrostatic field.
- Which radiation is defleced the most. Given reason.
- Which among the three radiations causes the least biological damage?
Explain why alpha and beta particles are deflected in an electric or a magnetic field, but gamma rays are not deflected in such a field.
Is it possible to deflect γ - radiations in a way similar to α and β -particles, using the electric or magnetic field? Give reasons.
State the speed of each of α, β and γ radiations.
What is the composition of α, β and γ radiations?
Arrange the α, β, and γ radiation in ascending order of their biological damage. Give reason.
Compare the penetrating powers of α, β and γ-radiations.
Compare the ionising powers of α, β and γ radiations.
The figure shows a radioactive source S placed in a thick-walled lead container. The radiations given off pass through a magnetic field acting in direction perpendicular to the plane of paper inwards as shown by X. Copy the diagram and show the path of radiations. Explain why the source is kept in a thick-walled lead container.