Advertisements
Advertisements
Question
Naphthalene can be separated from sand :
Options
by sublimation
by distillation
by crystallisation
by using water as solvent
Advertisements
Solution
by sublimation
Naphthalene sublimes on heating, whereas sand does not. So, when the mixture is heated, solid naphthalene turns into vapours, which can be condensed. Sand remains in the container.
APPEARS IN
RELATED QUESTIONS
Fill in the blank :
The state of matter with definite volume and definite shape is called______
Name two processes which provide the best evidence for the motion of particles in matter.
Describe in your own words, what happens to the particles when salt dissolves in water.
What name is given to those elements which are neither good conductors of electricity like copper nor insulators like sulphur ?
Which of the following are ‘pure substances’ ?
Ice, Milk, Iron, Hydrochloric acid, Calcium oxide, Mercury, Brick, Wood, Air
What are the three groups into which all the elements can be divided ? Name two elements belonging to each group.
Define a compound. Give two points of evidence to show that sodium chloride is a compound.
Name a metal which can be easily cut with a knife.
If 25 mL of acetone is present in 150 mL of its aqueous solution, calculate the concentration of solution.
Choose one term from the following which includes the other three :
aerosol, emulsion, colloid, sol
How will you differentiate between a suspension and a colloid ?
Name the process you would use to separate a mixture of water and alcohol.
Fill in the following blanks with suitable words :
Miscible liquids are separated by .....................
The correct increasing order of the boiling points of liquid oxygen, liquid argon and liquid nitrogen present in liquid air is :
Justify your answer.
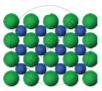
| Figure 1 | Figure 2 | Figure 3 |
 |
 |
 |
The arrangement of particles in three different phases of matter is shown above.
- Which state is represented by Fig. 1?
- In which state will the inter-particle attraction be maximum?
- Which one of them cannot be contained in an open vessel?
- Which one can take the shape of its container?
Why wires cannot be drawn from materials such as stone or wood?
Match the physical quantities given in column A to their S I units given in column B:
| (A) | (B) | ||
| (a) | Pressure | (i) | cubic metre |
| (b) | Temperature | (ii) | kilogram |
| (c) | Density | (iii) | pascal |
| (d) | Mass | (iv) | kelvin |
| (e) | Volume | (v) | kilogram per cubic metre |
Higher the temperature, the higher the kinetic energy of particles.
Gases do not diffuse very easily.
Write a scientific reason.
Lemon sherbat has sweet, sour and salty taste and it can be poured in a glass.
