Advertisements
Advertisements
Questions
Name the procedure used to prepare a sodium salt such as sodium sulphate.
Answer the following question related to salts and their preparations:
Name the procedure used to prepare a sodium salt such as sodium sulphate.
Advertisements
Solution 1
The name of the procedure used to prepare a sodium salt such as sodium sulphate is Neutralization of acid with base.
Solution 2
By neutralising alkali with acid. Pour the calculated 25 cm3 of NaOH into the flask. Using a pipette, add a few drops of phenolphthalein to it.
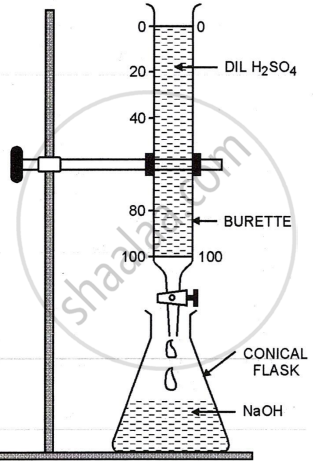
\[\ce{2NaOH + H2SO4 -> Na2SO4 + 2H2O}\]
Fill the burette with dil. H2SO4 upto zero mark from the burette add dil.H2SO4 drop by drop into the flask with constant stirring until pink colour just disappears. Transfer contents of flask to evaporating dish to evaporate water. On cooling crystals of [Na2SO4.10 H2O] are obtained.
\[\ce{Na2SO4 + 10H2O -> Na2SO4.10H2O}\]
APPEARS IN
RELATED QUESTIONS
How is the following salt prepared:
Sodium nitrate from sodium hydroxide.
Explain why fused calcium chloride is used in the preparation of FeCI3?
Explain hydronium ion. Write the ionization of sulphuric acid showing the formation of hydronium ion.
How many salts can be obtained from ortho phosphoric acid? Is there any difference in the salts formed by the acid?
From the list of the substances given, name the substances which you would use to prepare each of the following salts:
List of substances: Copper, lead, sodium, zinc, copper oxide, lead carbonate, sodium carbonate solution, dilute hydrochloric acid, dilute nitric acid and dilute sulphuric acid.
Salts: (a) zinc sulphate (b) copper sulphate (c) sodium sulphate (d) lead sulphate.
Write the balanced equation for the preparation of the following compounds, starting from iron and other substance:
lead sulphate using dilute sulphuric acid.
Give the equation for the preparation of the following salt from the starting material given.
Iron (III) Chloride from Iron
Explain the following method with example.
Direct combination
Fill in the blanks with suitable words:
An acid is a compound which when dissolved in water forms hydronium ions as the only ______ ions. A base is a compound which is soluble in water and contains ______ ions. A base reacts with an acid to form a ______ and water only. This type of reaction is known as ______.
Choose the most appropriate answer from
(SO2, SiO2, Al2O3, CO, MgO, Na2O)
An oxide which when dissolved in water form acid.
