Advertisements
Advertisements
Question
Justify that the following reaction is redox reaction; identify the species oxidized/reduced, which acts as an oxidant and which acts as a reductant.
\[\ce{HF_{(aq)} + {OH}^-_{ (aq)}->H2O_{(l)} + {F}^ -_{ (aq)}}\]
Advertisements
Solution
\[\ce{HF_{(aq)} + {OH}^-_{ (aq)}->H2O_{(l)} + {F}^ -_{ (aq)}}\]
- Write oxidation number of all the atoms of reactants and products
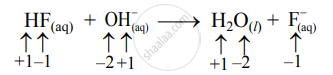
- Since the oxidation numbers of all the species remain the same, this is NOT a redox reaction.
Result:
The given reaction is NOT a redox reaction.
APPEARS IN
RELATED QUESTIONS
Choose the correct option.
Oxidation number of carbon in H2CO3 is
Choose the correct option.
Which of the following halogens does always show oxidation state -1?
In which chemical reaction does carbon exhibit variation of oxidation state from - 4 to + 4? Write a balanced chemical reaction.
In which reaction does nitrogen exhibit variation of oxidation state from –3 to +5?
Calculate the oxidation number of the underlined atom.
K2C2O4
Calculate the oxidation number of the underlined atom.
H2S4O6
Calculate the oxidation number of the underlined atom.
NaH2PO4
What is oxidation?
Identify the following pair of species is in its oxidized state.
Mg/Mg2+
Identify the following pair of species is in its oxidized state?
Cu/Cu2+
Provide the stock notation for the following compound:
Tl2O
Provide the stock notation for the following compound:
Fe2O3
Which of the following redox couple is a stronger oxidizing agent?
Cl2 (E0 = 1.36 V) and Br2 (E0 = 1.09 V)
Which of the following redox couple is a stronger reducing agent?
Li (E0 = - 3.05 V) and Mg (E0 = - 2.36 V)
Oxidation state of Xe in XeOF4 is ____________.
The oxidation number of sulphur in S8 molecule is ______.
Match the following.
| Compound | Oxidation no. of underlined element |
| i. \[\ce{\underline{C}_4H4O^{2-}_6}\] | a. +2.5 |
| ii. \[\ce{\underline{N}_3H}\] | b. +1.5 |
| iii. \[\ce{Mg2\underline{P}_2O7}\] | c. +5 |
| iv. \[\ce{Na2\underline{S}_4O6}\] | d. `-1//3` |
Oxidation state of nitrogen in nitric oxide is ______.
Which of the following is INCORRECT?
The sum of oxidation number of all atoms in \[\ce{SnO^{2-}_3}\] ion is _______.
Carbon is present in highest oxidation number in ______.
What is the oxidation number of Cr in K2Cr2O7?
What is the oxidation state of chlorine atom in perchloric acid?
The brown ring complex is formulated as [Fe(H2O)5NO]SO4. The oxidation number of iron is ______.
In which of the compounds does ‘manganese’ exhibit the highest oxidation number?
Find CORRECT statement for following reaction.
\[\ce{2Ag+ + Cu -> 2Ag + Cu^2+}\]
