Advertisements
Advertisements
Question
Indicate the complex ion which shows geometrical isomerism.
Options
\[\ce{[Cr(H2O)4Cl2]^+}\]
\[\ce{[Pt(NH3)3Cl]}\]
\[\ce{[Co(NH3)6]^{3+}}\]
\[\ce{[Co(CN)5(NC)]^{3-}}\]
Advertisements
Solution
\[\ce{[Cr(H2O)4Cl2]^+}\]
Explanation:
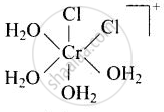
cis-isomer
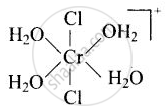
trans-isomer
APPEARS IN
RELATED QUESTIONS
Answer the following question.
Draw geometric isomers and enantiomers of the following complex.
[Pt(en)2ClBr]2⊕
Which one of the following complexes is not expected to exhibit isomerism?
Draw all possible geometrical isomers of the complex \[\ce{[Co(en)2Cl2]^+}\] and identify the optically active isomer.
Explain optical isomerism in coordination compounds with an example.
Consider the two complexes given below:
\[\ce{\underset{(I)}{[Co(NH3)5SO4]Br}}\] and \[\ce{\underset{(II)}{[Co(NH3)5Br]SO4}}\]
I and II are ____________ isomers.
Which of the following has zero dipole moment?
\[\ce{IUPAC}\] name of \[\ce{[Pt(NH3)2 Cl(NO2)]}\] is ______.
Geometrical isomerism is not shown by
Complex [COCl2(en)2]+ can
Which of the following compounds show optical isomerism?
Write the name of isomerism in the following complexes:
[Cu(NH3)4] [PtCl4] and [Pt(NH3)4] [ CuCl4]
The one that is not expected to show isomerism is ______.
Indicate the types of isomerism exhibited by the following complexes and draw the structures for these isomers: [Pt(NH3)(H2O)Cl2]
Write structures for geometrical isomers of diamminebromochloroplatinum (II).
Explain the geometrical isomerism of the octahedral complex of the type [M(AA)2B2]n± with a suitable example.
Draw the structure of trans isomers of Pt(NH3)2Cl2.
The co-ordination number of Co3+ ion in the complex [Co(NH3)4Cl2]⊕ is ______.
Which one of the following complex ions has geometrical isomers?
Match List-I with List-II:
| List-I (Complex) |
List-II (Type of isomerism) |
| A. [Co(NH3)5(NO2)]Cl2 | I. Solvate isomerism |
| B. [Co(NH3)5(SO4)]Br | II. Linkage isomerism |
| C. [Co(NH3)6][Cr(CN)6] | III. Ionisation isomerism |
| D. [Co(H2O)6]Cl3 | IV. Coordination isomerism |
Choose the correct answer from the options given below:
