Advertisements
Advertisements
Question
Draw all possible geometrical isomers of the complex \[\ce{[Co(en)2Cl2]^+}\] and identify the optically active isomer.
Advertisements
Solution
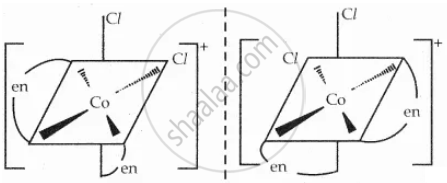
Two Cis \[\ce{[Co(en)2Cl2]^+}\] isomers - optically active
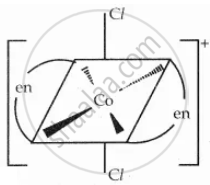
One trans \[\ce{[Co(en)2Cl2]^+}\] isomers - optically inactive
APPEARS IN
RELATED QUESTIONS
How many geometrical isomers are possible for \[\ce{[Pt(Py)(NH3)(Br)(Cl)]}\]?
What is linkage isomerism? Explain with an example.
The compound(s) that exhibit(s) geometrical isomerism is (are):
(I) [Pt(en)Cl2]
(II) [Pt(en)2]Cl2
(III) [Pt(en)2Cl2]
(IV) [Pt(NH3)2Cl2]
The complex ions [Co(H2O)5(ONO)]2+ and [Co(H2O)5NO2]2+ are ____________.
What type of isomerism is present between (I) [Cr(H2O)6]Cl3 and (II) [Cr(H2O)5Cl]Cl2.H2O?
Which of the following are isostructural pairs?
(A) \[\ce{SO^{2-}4}\] and \[\ce{CrO^{2-}4}\]
(B) SiCl4 and TiCl4
(C) NH3 and \[\ce{NO^-3}\]
(D) BCl3 and BrCl3
Give cis isomer of [Co(NH3)4Cl2]⊕.
Indicate the type of isomerism exhibited by the following complex and draw the structures for this isomer:
\[\ce{[Pt(NH3)(H2O)Cl2]}\]
Indicate the types of isomerism exhibited by the following complexes and draw the structures for these isomers: [Pt(NH3)(H2O)Cl2]
Give a chemical test to show that \[\ce{[Co(NH3)5Cl]SO4}\] and \[\ce{[Co(NH3)5SO4]CI}\] are ionisation isomers.
