Advertisements
Advertisements
प्रश्न
Draw all possible geometrical isomers of the complex \[\ce{[Co(en)2Cl2]^+}\] and identify the optically active isomer.
Advertisements
उत्तर
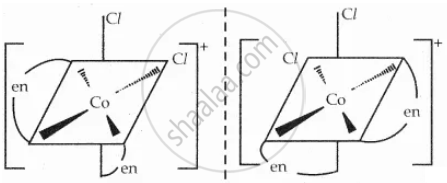
Two Cis \[\ce{[Co(en)2Cl2]^+}\] isomers - optically active
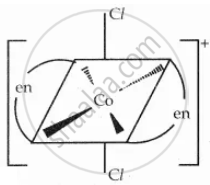
One trans \[\ce{[Co(en)2Cl2]^+}\] isomers - optically inactive
APPEARS IN
संबंधित प्रश्न
List various types of isomerism possible for coordination compounds, giving an example of each.
Which one of the following will give a pair of enantiomorphs?
Which kind of isomerism is possible for a complex [Co(NH3)4Br2]Cl?
The term anomers of glucose refer to ____________.
Which would exhibit coordination isomerism?
Which of the following is NOT a pair of structural isomers?
Which of the following does NOT show optical isomerism?
What type of isomerism is present between (I) [Cr(H2O)6]Cl3 and (II) [Cr(H2O)5Cl]Cl2.H2O?
Indicate the type of isomerism exhibited by the following complex and draw the structures for this isomer:
\[\ce{[Pt(NH3)(H2O)Cl2]}\]
Explain the geometrical isomerism of the octahedral complex of the type [M(AA)2B2]n± with a suitable example.
