Advertisements
Advertisements
प्रश्न
Draw all possible geometrical isomers of the complex \[\ce{[Co(en)2Cl2]^+}\] and identify the optically active isomer.
Advertisements
उत्तर
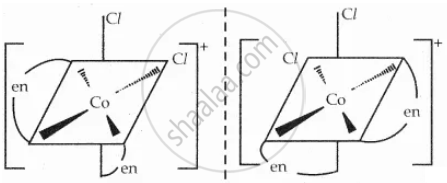
Two Cis \[\ce{[Co(en)2Cl2]^+}\] isomers - optically active
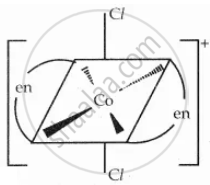
One trans \[\ce{[Co(en)2Cl2]^+}\] isomers - optically inactive
APPEARS IN
संबंधित प्रश्न
The pair [Co(NH3)5(SO4)]Br and [Co(NH3)5Br]SO4 exhibits ____________ isomerism
Draw optical isomers of [Co(en)3]3+.
The number of geometrical isomers of [CrCl2(en)2]+ is ____________.
____________ isomers are formed when the ligand has two different donor atoms.
What is the number of moles of silver chloride precipitated when excess of aqueous silver nitrate is treated with [Co(NH3)4Cl2]Cl?
Indicate the complex ion which shows geometrical isomerism.
Geometrical isomerism is not shown by
Which of the following are isostructural pairs?
(A) \[\ce{SO^{2-}4}\] and \[\ce{CrO^{2-}4}\]
(B) SiCl4 and TiCl4
(C) NH3 and \[\ce{NO^-3}\]
(D) BCl3 and BrCl3
White precipitate of AgCl dissolves in aqueous ammonia solution due to formation of ______.
Match List-I with List-II:
| List-I (Complex) |
List-II (Type of isomerism) |
| A. [Co(NH3)5(NO2)]Cl2 | I. Solvate isomerism |
| B. [Co(NH3)5(SO4)]Br | II. Linkage isomerism |
| C. [Co(NH3)6][Cr(CN)6] | III. Ionisation isomerism |
| D. [Co(H2O)6]Cl3 | IV. Coordination isomerism |
Choose the correct answer from the options given below:
