Advertisements
Advertisements
प्रश्न
Indicate the complex ion which shows geometrical isomerism.
विकल्प
\[\ce{[Cr(H2O)4Cl2]^+}\]
\[\ce{[Pt(NH3)3Cl]}\]
\[\ce{[Co(NH3)6]^{3+}}\]
\[\ce{[Co(CN)5(NC)]^{3-}}\]
Advertisements
उत्तर
\[\ce{[Cr(H2O)4Cl2]^+}\]
Explanation:
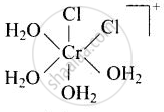
cis-isomer
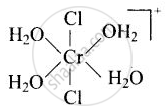
trans-isomer
APPEARS IN
संबंधित प्रश्न
List various types of isomerism possible for coordination compounds, giving an example of each.
Define the term Co-ordination isomer.
Which one of the following complexes is not expected to exhibit isomerism?
Fac-mer isomerism is shown by
Draw all possible geometrical isomers of the complex \[\ce{[Co(en)2Cl2]^+}\] and identify the optically active isomer.
Which would exhibit coordination isomerism?
Which of the following does NOT show optical isomerism?
How many donor groups are present in diethylene triamine?
Complex [COCl2(en)2]+ can
Which of the following compounds show optical isomerism?
Which of the following shows maximum number of isomers?
White precipitate of AgCl dissolves in aqueous ammonia solution due to formation of ______.
Which among the following solid is a non-polar solid?
Indicate the type of isomerism exhibited by the following complex and draw the structures for this isomer:
\[\ce{[Pt(NH3)(H2O)Cl2]}\]
Explain the ionisation isomers.
Draw the structure of cis isomers of Pt(NH3)2Cl2.
Name the type of isomerism exhibited by the following pair of compounds:
[Co(NH3)5(ONO)]Cl2 and [Co(NH3)5(NO2)]Cl2
Match List-I with List-II:
| List-I (Complex) |
List-II (Type of isomerism) |
| A. [Co(NH3)5(NO2)]Cl2 | I. Solvate isomerism |
| B. [Co(NH3)5(SO4)]Br | II. Linkage isomerism |
| C. [Co(NH3)6][Cr(CN)6] | III. Ionisation isomerism |
| D. [Co(H2O)6]Cl3 | IV. Coordination isomerism |
Choose the correct answer from the options given below:
