Advertisements
Advertisements
Question
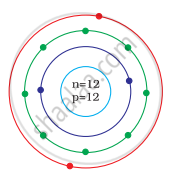
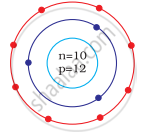
Identify the Mg2+ ion from the Fig. where, n and p represent the number of neutrons and protons respectively
Options
Advertisements
Solution

Explanation -
In magnesium atom `""_12^24"Mg"`
Number of n = 12, p = 12, e = 12
Number of electrons in Mg2+ = 10
APPEARS IN
RELATED QUESTIONS
Name the three sub-atomic particles of an atom.
Compare the properties of electrons, protons and neutrons.
The mass of an electron is about `1/2000` times that of proton.
Which subatomic particle is not present in an ordinary hydrogen atom?
Name the subatomic particle whose relative charge is : 0
Calculate the number of neutrons, protons and electrons:
- atomic number 3 and mass number 7
- atomic number 92 and mass number 238.
On the basis of Rutherford’s model of an atom, which subatomic particle is present in the nucleus of an atom?
Calculate the number of neutrons present in the nucleus of an element X which is represented as `""_15^31"X"`
Write the properties of neutrons.
The ratio of neutrons in C and Si with respective atomic masses 12 and 28 is ______.