Advertisements
Advertisements
प्रश्न
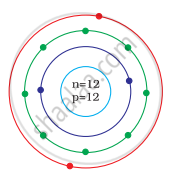
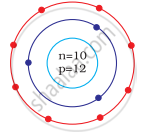
Identify the Mg2+ ion from the Fig. where, n and p represent the number of neutrons and protons respectively
पर्याय
Advertisements
उत्तर

Explanation -
In magnesium atom `""_12^24"Mg"`
Number of n = 12, p = 12, e = 12
Number of electrons in Mg2+ = 10
APPEARS IN
संबंधित प्रश्न
Name the three sub-atomic particles of an atom.
Name the subatomic particle whose relative charge is : 0
Explain in brief the experimental proof which led to the discovery of –
Neutrons
State the number of neutrons in each of the atoms A to E. Also state which of the atoms A to E is a metal.

On the basis of Rutherford’s model of an atom, which subatomic particle is present in the nucleus of an atom?
Calculate the number of neutrons present in the nucleus of an element X which is represented as `""_15^31"X"`
The hydrogen atom does not have ______.
α, β, γ rays are during the ratio active decay ______ of an atom.
Beryllium + α – rays → Carbon + ______.
The stability of the nucleus is determined by the Neutron – Proton ratio.