Advertisements
Advertisements
प्रश्न
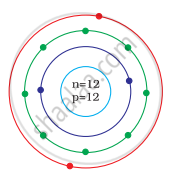
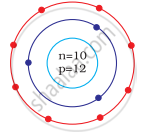
Identify the Mg2+ ion from the Fig. where, n and p represent the number of neutrons and protons respectively
विकल्प
Advertisements
उत्तर

Explanation -
In magnesium atom `""_12^24"Mg"`
Number of n = 12, p = 12, e = 12
Number of electrons in Mg2+ = 10
APPEARS IN
संबंधित प्रश्न
Helium atom has an atomic mass of 4 u and two protons in its nucleus. How many neutrons does it have?
Which subatomic particle is not present in an ordinary hydrogen atom?
Which subatomic particle was discovered by : Chadwick ?
Fill in the blank of the following statement :
If the nucleus of an atom has atomic number 17, mass number 37 and there are 17 electrons outside the nucleus, the number of neutrons in it is __________.
Isotopes exist because atoms of the same element can have different numbers of ______.
What is the charge of a neutron?
Write the properties of neutrons.
The hydrogen atom does not have ______.
Which subatomic particles have no charge?
The stability of the nucleus is determined by the Neutron – Proton ratio.