Advertisements
Advertisements
Question
How will you indicate a solution made in water in a chemical equation?
Advertisements
Solution
A solution made in water is indicated by writing (aq) after its formula in a chemical equation.
Example: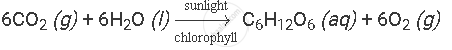
APPEARS IN
RELATED QUESTIONS
What is a balanced chemical equation? Why should a chemical equation be balanced?
Write complete balanced equation for the following reaction:
Sulphur dioxide (gas) + Oxgyen (gas)  Sulphur trioxide (gas)
Sulphur trioxide (gas)
The chemical equations are balanced to satisfy one of the following laws in chemical reactions. This law is know as:
(a) law of conservation of momentum
(b) law of conservation of mass
(c) law of conservation of motion
(d) law of conservation of magnetism
Name the type of chemical reaction shown by the following equation:
Fe + CuSO4 → FeSO4 +Cu
Write word equation for the following skeletal equation:
\[\ce{AgBr -> Ag + Br2}\]
Write symbolic representation for the following word equation and balance them :
Calcium carbonate → Calcium oxide + Carbon dioxide.
Balance the following equation. Also name the products formed.
\[\ce{Ag + S -> Ag2S}\]
Balance the equation stepwise.
SO2(g) + H2S(aq) → S(s) + H2O(l)
State the information conveyed by the following equation:
Zn(s) + 2HCl(aq) → ZnCl2(aq) + H2 ↑
Balance the following equation:
Pb3O4 → PbO + O2
Write the balanced chemical equation of the following reaction.
aluminium carbide + water → aluminium hydroxide + methane
Write word equation for the following molecular equation:
\[\ce{CaCO3 + 2HCl ->[\triangle] CaCl2 + H2O + CO2 [g]}\]
Word equation:
State which characteristic does [g] in the above reaction indicate. The word equation of:
Write word equation for the following molecular equation:
FeSO4 + 2NaOH →Na2SO4 + Fe(OH)2 ↓
Word equation:
State why [↓] indication is seen after Fe(OH)2 and not after Na2SO4
Give one example in the case where supplying energy [given below] is necessary for a chemical reaction.
Pressure
Balance the following simple equation:
Mg + N2 → Mg3N2
Balance the following simple equation:
Al + N2 → AlN
Balance the following chemical equation.
- Na + O2 → Na2O3
- Ca + N2 → Ca3N2
- N2 + H2 →NH3
- CaCO3 +HCl → CaCl2 + CO2 +H2O
- Pb(NO3)2 → PbO + NO2 + O2
Which of the following are exothermic processes?
- Reaction of water with quick lime
- Dilution of an acid
- Evaporation of water
- Sublimation of camphor (crystals)
Complete the following blank in the equation as indicated.
\[\ce{CaH2_{(s)} + 2H2O_{( aq)}-> Ca(OH)2_{(s)} + 2H2_{(g)}}\]
Grams: 42 g + ______ `→` ______ + ______
