Advertisements
Advertisements
प्रश्न
How will you indicate a solution made in water in a chemical equation?
Advertisements
उत्तर
A solution made in water is indicated by writing (aq) after its formula in a chemical equation.
Example: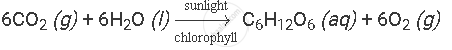
APPEARS IN
संबंधित प्रश्न
What is wrong with the following chemical equation?
Mg + O → MgO
Correct and balance it.
Balance the given equation:
BaCI2 + H2SO4  BaSO4 + HCI
BaSO4 + HCI
What are the various ways in which a chemical equation can be made more informative? Give examples to illustrate your answer.
Explain, with example, how the physical states of the reactants and products can be shown in a chemical equation.
Aluminium hydroxide reacts with sulphuric acid to form aluminium sulphate and water. Write a balanced equation for this reaction.
You are given the solution of lead nitrate. In order to obtain a yellow precipitate you should mix with it a solution of:
(a) potassium chloride
(b) potassium nitride
(c) potassium sulphide
(d) potassium iodide
Write the balanced equation for the following chemical reaction.
\[\ce{Barium chloride + Aluminium sulphate -> Barium sulphate + Aluminium chloride}\]
Write your observation for the following chemical reaction and name the product formed :
When dilute acetic acid is poured on baking soda.
Balance the following equation :
Fe + O2 → Fe3 O4
Balance the following equation. Also name the product formed.
FeCI2 + CI2 → FeCI3
Write the chemical equation for the following word equation and balance them.
Magnesium + Sulphuric acid → Magnesium sulphate + Hydrogen
Balance the following equation:
NaHCO3 → Na2CO3 + H2O + CO2
Write the balanced chemical equation of the following reaction.
potassium bicarbonate + sulphuric acid → potassium sulphate + carbon dioxide + water
Write the balanced chemical equation of the following reaction.
Potassium permanganate + hydrochloric acid → potassium chloride + manganese chloride + chlorine + water
Grills of doors and windows are always painted before they are used.
Write a balanced equation for the following word equation:
Potassium bromide + Chlorine → Potassium chloride + Bromine
Give a balanced equation by partial equation method, [steps are given below].
The reaction of excess ammonia with chlorine – Ammonia as a reducing agent
- Ammonia first reacts with chlorine to give hydrogen chloride and nitrogen.
- Hydrogen chloride then further reacts with excess ammonia to give ammonium chloride.
Give a balanced equation by the partial equation method, [steps are given below].
Oxidation of Lead [II] Sulphide by Ozone
- Ozone first decomposes to give molecular oxygen & nascent oxygen.
- Nascent oxygen then oxidises lead [II] sulphide to lead [II] sulphate.
Underline the compound in the equation given below, it is incorrectly balanced and write the correct balancing for the same.
Cl2 + 2H2O + SO2 → 4HCl + H2SO4
Complete the following blank in the equation as indicated.
\[\ce{CaH2_{(s)} + 2H2O_{( aq)}-> Ca(OH)2_{(s)} + 2H2_{(g)}}\]
Molecules: 6.02 × 1023 + ______ `→` ______ + ______
