Advertisements
Advertisements
प्रश्न
How will you indicate a solution made in water in a chemical equation?
Advertisements
उत्तर
A solution made in water is indicated by writing (aq) after its formula in a chemical equation.
Example: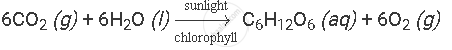
APPEARS IN
संबंधित प्रश्न
Translate the following statement into chemical equation and then balance the equation:
Phosphorus burns in oxygen to give phosphorus pentoxide.
Write complete balanced equation for the following reaction:
Calcium (solid) + water (liquid)  Calcium Hydroxide (solution) + Hydrogen (gas)
Calcium Hydroxide (solution) + Hydrogen (gas)
When hydrogen is passed over copper oxide, copper and steam are formed. Write a balanced equation for this reaction and state which of the chemicals are reactants.
Explain, with example, how the physical states of the reactants and products can be shown in a chemical equation.
With the help of an appropriate example, justify that some of the chemical reactions are determined by Change in colour
Give chemical equation for the reaction involved in the above case.
What do you understand by exothermic reactions?
Balance the equation stepwise.
H2S2O7(l) + H2O(l) → H2SO4(l)
Write the chemical equation for the following word equation and balance them.
Nitrogen + Oxygen → Nitrogen monoxide
Write the chemical equation for the following word equation and balance them.
Sodium reacts with water to form sodium hydroxide and hydrogen
Balance the following equation:
Fe + H2O → Fe3O4 + H2
Balance the following equation:
Ca + N2 → Ca3N2
Balance the following equation:
PbO + NH3 → Pb + H2O + N2
Balance the following equation:
Pb3O4 → PbO + O2
\[\ce{MnO2 + 4HCl -> MnCl2 + 2H2O + Cl2}\]
0.02 moles of pure MnO2 is heated strongly with conc. HCl. Calculate moles of acid required.
A chemical reaction is generally accompanied by certain external indications or characteristics. These include – change of – (a) colour (b) state (c) smell (d) evolution of gas (e) formation of precipitate (f) evolution or absorption of heat. With reference to change of colour – state the change in colour seen when the following are heated – copper carbonate.
Representation of the results of a chemical change – is a chemical equation.
For the equation: FeCl3 + 3NH4OH 3NH4Cl + Fe(OH)3 ↓
Answer the following:
Name the reactants and the products in the above equation.
Balance the following simple equation:
NaHCO3 + H2SO4 → Na2SO4 + H2O + CO2
CaCO3 + 2HCl[dil.] → CaCl2 + H2O + CO2 [g]
State the information not conveyed by the above chemical equation.
What is a chemical equation?
