Advertisements
Advertisements
Question
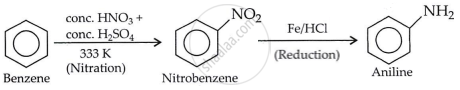
How will you convert Benzene into aniline?
Advertisements
Solution
APPEARS IN
RELATED QUESTIONS
Arrange the following: C2H5NH2, C2H5OH, (CH3)3N – in the increasing order of their boiling point
Arrange the following in increasing order of their basic strength :

- Write structures of different isomeric amines corresponding to the molecular formula C4H11N.
- Write the IUPAC names of all the isomers.
- What type of isomerism is exhibited by different pairs of amines?
Arrange the following in increasing order of their basic strength:
C2H5NH2, (C2H5)2NH, (C2H5)3N, C6H5NH2
Arrange the following in increasing order of their basic strength:
CH3NH2, (CH3)2NH, (CH3)3N, C6H5NH2, C6H5CH2NH2
Complete the following acid-base reaction and name the product:
\[\ce{(C2H5)3N + HCl ->}\]
Arrange the following:
In increasing order of boiling point:
C2H5OH, (CH3)2NH, C2H5NH2
Accomplish the following conversion:
Benzene to m-bromophenol
Accomplish the following conversion:
Aniline to p-bromoaniline
Give reasons CH3NH2 is more basic than C6H5NH2.
Give reason (CH3)2NH is more basic than (CH3)3N in an aqueous solution.
Arrange the following in the increasing order of their pKb values:
C6H5NH2, C2H5NH2, C6H5NHCH3
Arrange the following in increasing order of basic strength :
C6H5NH2, C6H5NHCH3, C6H5N(CH3)2
Choose the most correct option.
Which one of the following compounds has the highest boiling point?
Account for the following:
N-ethylethanamine boils at 329.3 K and butanamine boils at 350.8 K, although both are isomeric in nature.
Acetic acid exist as dimer in benzene due to
The hydrogen bond is shortest in
Which of the following amines form maximum hydrogen bonds within themselves?
