Advertisements
Advertisements
Question
How do fishes and aquatic animals survive when the pond gets covered with thick ice?
Advertisements
Solution
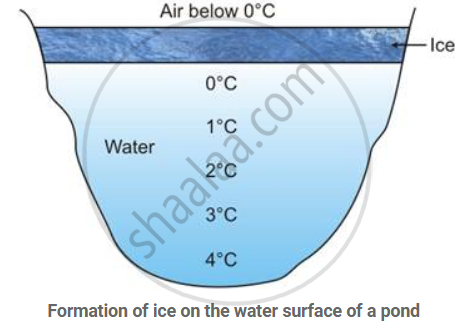
Water has an unusual physical property. When cooled, it first contracts in volume, as do other liquids, but at 4°C (maximum density), it starts expanding, and continues to do so till the temperature reaches 0°C, the point at which it freezes into ice.
The property of anomalous expansion of water enables marine life to exist in the colder regions of the world, because even when the water freezes on the surface, it is still liquid below the ice layer.
APPEARS IN
RELATED QUESTIONS
What is the smallest particle of water ? Describe it’s structure.
Explain how the peculiar variation of density of water with temperature can cause water— pipes to burst in very cold places.
How are clouds formed?
Explain why:
Rain water does not leave behind concentric rings when boiled.
Define ‘eutrophication’.
Match the following
| 1. | Flood | Lake |
| 2. | Surface water | Evaporation |
| 3. | Sunlight | Water vapour |
| 4. | Cloud | Pole |
| 5. | Frozen water | Increased rainfall |
Look at the map of Tamilnadu showing annual rainfall and answer the questions given below

- Identify the districts that get only low annual rainfall in Tamilnadu.
- Identify the districts that get a medium annual rainfall in Tamilnadu.
- State the districts that enjoy high annual rainfall in Tamilnadu.
Water is colourless, odourless and _______.
______ plays a vital role in the evolution and survival of life.
A mixture (x) in water contains suspended solids, organic impurities, inorganic impurities (a), nutrients (b), disease-causing bacteria and other microbes. Give names for (x), (a) and (b)?
