Advertisements
Advertisements
प्रश्न
How do fishes and aquatic animals survive when the pond gets covered with thick ice?
Advertisements
उत्तर
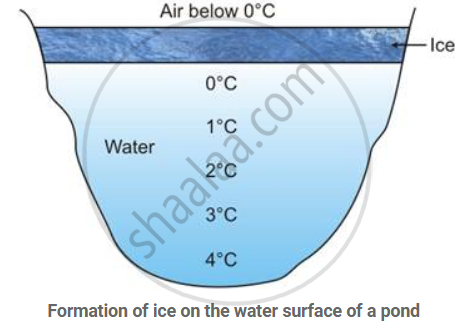
Water has an unusual physical property. When cooled, it first contracts in volume, as do other liquids, but at 4°C (maximum density), it starts expanding, and continues to do so till the temperature reaches 0°C, the point at which it freezes into ice.
The property of anomalous expansion of water enables marine life to exist in the colder regions of the world, because even when the water freezes on the surface, it is still liquid below the ice layer.
APPEARS IN
संबंधित प्रश्न
Name the four main sources of water.
Give balanced chemical equation for the reaction of water with Sodium .
Name the gas produced when chlorine is dissolved in water and exposed to sunlight.
Give reason of Carbonated drinks are bottled under high pressure.
Give one word for the following statement:
The harmful substances dissolved in water .
How are natural springs of water formed ?
How is aquatic life benefited by the fact that water has maximum density at 4oC?
Name a deliquescent substance’.
Name the process which is responsible for the formation of droplets on the surface of a glass of cold drink. _______
Write a few slogans of your own on the topic “Save Water”
