Advertisements
Advertisements
Question
Given the following data for incident wavelength and the stopping potential obtained from an experiment on the photoelectric effect, estimate the value of Planck's constant and the work function of the cathode material. What is the threshold frequency and corresponding wavelength? What is the most likely metal used for emitter?
| Incident wavelength (in Å) | 2536 | 3650 |
| Stopping potential (in V) |
1.95 | 0.5 |
Advertisements
Solution
Data: λ = 2536 Å = 2.536 × 10-7 m,
λ' = 3650 Å = 3.650 × 10-7 m,
V0 = 1.95 V, V0' = 0.5 V, c = 3 × 108 m/s,
e = 1.6 × 10-19 C
(i) `"V"_0"e" = "hc"/lambda - phi and "V"_0'"e" = "hc"/(lambda') - phi`
∴ `("V"_0 - "V"_0')"e" = "hc" (1/lambda - 1/(lambda '))`
∴ (1.95 - 0.5)(1.6 × 10-19)
= h (3 × 108)`(10^7/2.536 - 10^7/3.650)`
∴ 2.32 × 10-19 = h(3 × 1015)(0.3943 - 0.2740)
∴ h = `(2.32 xx 10^(-34))/0.3609 = 6.428 xx 10^(-34)` J.s
This is the value of Planck's constant.
(ii) `phi = "hc"/lambda - "V"_0"e"`
`= ((6.428 xx 10^-34)(3 xx 10^8))/(2.536 xx 10^-7) - (1.95)(1.6 xx 10^-19)`
`= 7.604 xx 10^-19 - 3.12 xx 10^-19 = 4.484 xx 10^-19`J
`= (4.484 xx 10^-19"J")/(1.6 xx 10^-19 "J"//"eV")`
= 2.803 eV
This the work function of the cathode material.
(iii) Φ = hv0
∴ The threshold frequency, `"v"_0 = phi/"h"`
`= (4.484 xx 10^-19 "J")/(6.428 xx 10^-34 "J.s") = 6.976 xx 10^14` Hz
(iv) `"v"_0 = "c"/lambda_0`
∴ The threshold wavelength, `lambda_0 = "c"/"v"_0`
`= (3 xx 10^8)/(6.976 xx 10^14) = 4.300 xx 10^-7` = 4300 Å
(v) The most likely metal used for emitter: calcium.
APPEARS IN
RELATED QUESTIONS
Choose the correct option.
Polychromatic (containing many different frequencies) radiation is used in an experiment on the photoelectric effect. The stopping potential ______.
Radiation of wavelength 4500 Å is incident on a metal having work function 2.0 eV. Due to the presence of a magnetic field B, the most energetic photoelectrons emitted in a direction perpendicular to the field move along a circular path of radius 20 cm. What is the value of the magnetic field B?
The minimum frequency for photoelectric effect on metal is 7 × 1014 Hz, Find the work function of the metal.
With the help of a circuit diagram describing an experiment to study the photoelectric effect.
What is the photoelectric effect? Define stopping potential and photoelectric work function.
The energy of a photon is 2 eV. Find its frequency and wavelength.
The maximum velocity of photoelectron emitted is 4.8 m/s. If the e/m ratio of the electron is 1.76 × 1011 C/kg, then stopping potential is given by ______
When a photon enters glass from air, which one of the following quantity does not change?
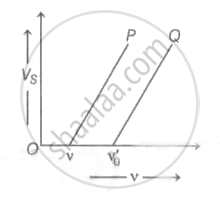
The graph of stopping potential `"V"_"s"` against frequency v of incident radiation is plotted for two different metals P and Q as shown in the graph. ΦP and ΦQ are work-functions of P and Q respectively, then
Threshold frequency for a metal is 1015 Hz. Light of `lambda` = 4000 Å falls on its surface. Which of the following statements is correct?
Light of wavelength `lambda` strikes a photo-sensitive surface and electrons are ejected with kinetic energy E. If the kinetic energy is to be increased to 2E, the wavelength must be changed to `lambda'` where ____________.
Threshold wavelength for lithium metal is 6250 Å. For photoemission, the wavelength of the incident light must be ______.
The threshold frequency for a certain photosensitive metal is v0. When it is illuminated by light of frequency v = 2v0, the maximum velocity of photoelectrons is v0. What will be the maximum velocity of the photoelectrons when the same metal is illuminated by light of frequency
v = 5v0?
The work function of a substance is 4.0 eV. The longest wavelength of light that can cause photo-emission from this substance is approximately (h = 6.63 × 10-34 Js)[1eV = 1.6 × 10-19 J]
The photon of frequency vis incident on a metal surface whose threshold frequency is v0. The kinetic energy of the emitted photoelectrons will be ____________.
The photon of frequency vis incident on a metal surface whose threshold frequency is v0. The kinetic energy of the emitted photoelectrons will be ______.
The work function of a photosensitive material is 4.0 eV. The longest wavelength of light that can cause photon emission from the substance is (approximately) ____________.
An electromagnetic wave of wavelength '`lambda`' is incident on a photosensitive surface of negligible work function. If the photoelectrons emitted from this surface have the de-Broglie wavelength '`lambda_1`' then ____________.
The radiation corresponding to the 3 → 2 transition of a hydrogen atom falls on a gold surface to generate photoelectrons. These electrons are passed through a magnetic field of 5 × 10-4 T. Assume that the radius of the largest circular path followed by these electrons is 7 mm, and the work function of the metal is ______.
(Mass of electron = 9.1 × 10-31 kg)
In a photoelectric experiment, ultraviolet light of wavelength 280 nm is used with a lithium cathode having work function Φ = 2.5 eV. If the wavelength of incident light is switched to 400 nm, find out the change in the stopping potential.
(h = 6.63 × 10-34 Js, c = 3 × 108 ms-1)
We wish to observe an object which is 2.5Å in size. The minimum energy photon that can be used ______.
If the electron in hydrogen atom jumps from second Bohr orbit to ground state and difference between energies of the two states is radiated in the form of photons. If the work function of the material is 4.2 eV, then stopping potential is ______.
[Energy of electron in nth orbit = `-13.6/"n"^2` eV ]
On a photosensitive material when frequency of incident radiation is increased by 30%, kinetic energy of emitted photoelectrons increases from 0.4 eV. The work function of the surface is ______.
Light of wavelength λ, which is less than threshold wavelength is incident on a photosensitive material. If incident wavelength is decreased so that emitted photoelectrons are moving with same velocity, then stopping potential will ______.
Photoelectric emission is observed from a metallic surface for frequencies ν1 and ν2 of the incident light rays (ν1 > ν2). If the ratio of the maximum value of the kinetic energy of the photoelectrons emitted in the first case to that in the second case is 2 : K, then the threshold frequency of the metallic surface is ______.
The photoelectric threshold for a certain metal surface is 3600 Å. If the metal surface is irradiated by a wavelength of 1100 Å, then kinetic energy of the emitted photoelectrons is ______.
Explain the failure of wave theory of light to account for the observations from experiments on photoelectric effect.
Give Einstein's explanation of the photoelectric effect.
Draw a neat labelled diagram of photo-current as a function of accelerating potential for fixed incident intensity but different incident frequencies for the same emitter material.
Explain the formation of clouds at high altitude.
