Advertisements
Advertisements
Question
Choose the correct option.
Polychromatic (containing many different frequencies) radiation is used in an experiment on the photoelectric effect. The stopping potential ______.
Options
Will depend on the average wavelength
Will depend on the longest wavelength
Will depend on the shortest wavelength
Does not depend on the wavelength
Advertisements
Solution
Polychromatic (containing many different frequencies) radiation is used in an experiment on the photoelectric effect. The stopping potential will depend on the shortest wavelength.
Explanation:
The photoelectric effect is the phenomenon of emission of the electron when the photon incident on the metallic surfaces.
• The electrons emitted from the metallic surface due to the photoelectric effect are called photoelectrons.
• The rate of emission of photoelectrons depends upon the frequency or wavelength of the incident light.
• When higher frequency or lower wavelength light incident on the metallic surface, more photoelectrons will be emitted giving rise to the photocurrent.
• Stopping potential is the minimum potential required for the prevention of ejection of photoelectrons due to the low wavelength radiations.
• In the case of polychromatic light, the radiation with high frequency and low wavelength will contribute to the ejection of electrons.
APPEARS IN
RELATED QUESTIONS
Can microwaves be used in the experiment on photoelectric effect?
The threshold wavelength of tungsten is 2.76 x 10-5 cm.
(a) Explain why no photoelectrons are emitted when the wavelength is more than 2.76 x 10-5 cm.
(b) What will be the maximum kinetic energy of electrons ejected in each of the following cases
(i) if ultraviolet radiation of wavelength λ = 1.80 × 10-5 cm and
(ii) radiation of frequency 4 x 1015 Hz is made incident on the tungsten surface?
Define photoelectric effect.
Draw a neat labelled diagram of a schematic of the experimental setup for the photoelectric effect.
Explain the concept of the photoelectric effect.
If the total energy of radiation of frequency 1014 Hz is 6.63 J, Calculate the number of photons in the radiation.
With the help of a circuit diagram describing an experiment to study the photoelectric effect.
State Einstein’s photoelectric equation. Explain all characteristics of the photoelectric effect, on the basis of Einstein’s photoelectric equation.
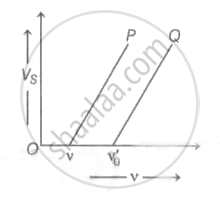
The graph of stopping potential `"V"_"s"` against frequency v of incident radiation is plotted for two different metals P and Q as shown in the graph. ΦP and ΦQ are work-functions of P and Q respectively, then
When light falls on a metal surface, the maximum kinetic energy of the emitted photoelectrons depends upon ______
Threshold wavelength for lithium metal is 6250 Å. For photoemission, the wavelength of the incident light must be ______.
When certain metal surface is illuminated with a light of wavelength A., the stopping potential is V, When the same surface is illuminated by light of wavelength 2λ, the stopping potential is `("V"/3)`. The threshold wavelength for the surface is ______.
When wavelength of incident radiation on the metal surface is reduced from 'λ1' to 'λ2', the kinetic energy of emitted photoelectrons is tripled. The work function of the metal is ______.
(h = Planck's constant, c =velocity of light)
When light of wavelength 'λ' is incident on a photosensitive surface, the stopping potential is 'V'. When light of wavelength '3λ' is incident on the same surface, the stopping potential is `"V"/6`. Threshold wavelength for the surface is _______.
Light of different frequencies, whose photons have energies 3 eV and 18 eV respectively, successively illuminate a metal of work function 2 eV. The ratio of the maximum speeds of the emitted electrons will be ______.
The work function of a substance is 4.0 eV. The longest wavelength of light that can cause photo-emission from this substance is approximately (h = 6.63 × 10-34 Js)[1eV = 1.6 × 10-19 J]
The photon of frequency vis incident on a metal surface whose threshold frequency is v0. The kinetic energy of the emitted photoelectrons will be ____________.
The photon of frequency vis incident on a metal surface whose threshold frequency is v0. The kinetic energy of the emitted photoelectrons will be ______.
When a metal with work function 0.6 eV is illuminated with light of energy 2 eV, the stopping potential will be ____________.
An electromagnetic wave of wavelength '`lambda`' is incident on a photosensitive surface of negligible work function. If the photoelectrons emitted from this surface have the de-Broglie wavelength '`lambda_1`' then ____________.
When light of wavelength '`lambda`' is incident on photosensitive surface, photons of power 'P' are emitted. The number of photons (n) emitted in 't' second is (h = Planck's constant, c = velocity of light in vacuum) ____________.
The stopping potential in the context of photoelectric effect depends on the following property of incident electromagnetic radiation ______.
In a photoelectric experiment, ultraviolet light of wavelength 280 nm is used with a lithium cathode having work function Φ = 2.5 eV. If the wavelength of incident light is switched to 400 nm, find out the change in the stopping potential.
(h = 6.63 × 10-34 Js, c = 3 × 108 ms-1)
The maximum kinetic energy of the photoelectrons ejected will be ______ eV when the light of wavelength 350 nm is incident on a cesium surface. The work function of cesium = 1.9 eV.
A charged dust particle of radius 5 × 10-7 m is located in a horizontal electric field having an intensity of 6.28 × 105 V/m. The surrounding medium is air with a coefficient of viscosity η = 1.6 × 10-5 N-s/m2. If the particle moves with a uniform horizontal speed of 0.02 m/s, the number of electrons on it is ______.
The following graphs show the variation of stopping potential corresponding to the frequency of incident radiation (ν) for a given metal. The correct variation is shown in graph [ν0 = threshold frequency].
|
(A) |
(B) |
|
(C) |
(D) |
When monochromatic light of frequency v1 falls on a metal surface, the stopping potential required is found to be V1. If the radiation of frequency v2 is incident on the surface, the stopping potential required V2 is ______. (v2 > v1)




