Advertisements
Advertisements
Question
Give the postulates of Bohr's atomic model
Advertisements
Solution
Atom has a central nucleus surrounded by electrons.
The electrons revolve around the nucleus in definite circular paths called orbits.
Each orbit has fixed energy. Therefore, these orbits are also known as energy levels or energy shells.
These orbits or energy levels are represented either by a number 1, 2, 3,... known as the principal quantum number (n) of the orbit or by K, L, M,....
There is no change of energy of electrons as long as they keep revolving in the same energy level and the atom remains stable. Electrons can jump from the higher orbit to the lower orbit when they lose energy and jump to the next when they gain energy.
APPEARS IN
RELATED QUESTIONS
Draw a sketch of Bohr’s model of an atom with three shells.
Name the scientists who described the arrangement of electrons in an atom.
Describe Bohr's model of the atom. How did Neils Bohr explain the stability of atom?
State in brief the drawbacks of Rutherford's atomic model correlating them with the postulates of Bohr’s atomic model.
Explain the postulates of Bohr’s atomic model.
An atom with 3 protons and 4 neutrons will have a valency of
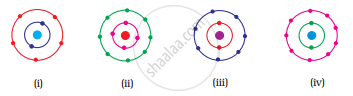
Which of the following in Fig. 4.2 do not represent Bohr’s model of an atom correctly?
Niels Bohr was Born on ______.
Bohr's model of atoms ______.
The angular momentum of electron in hydrogen atom in first orbit is ‘L’. The change in angular momentum if electron is in second orbit of hydrogen atom is ______.
