Advertisements
Advertisements
प्रश्न
Give the postulates of Bohr's atomic model
Advertisements
उत्तर
Atom has a central nucleus surrounded by electrons.
The electrons revolve around the nucleus in definite circular paths called orbits.
Each orbit has fixed energy. Therefore, these orbits are also known as energy levels or energy shells.
These orbits or energy levels are represented either by a number 1, 2, 3,... known as the principal quantum number (n) of the orbit or by K, L, M,....
There is no change of energy of electrons as long as they keep revolving in the same energy level and the atom remains stable. Electrons can jump from the higher orbit to the lower orbit when they lose energy and jump to the next when they gain energy.
APPEARS IN
संबंधित प्रश्न
Draw a sketch of Bohr’s model of an atom with three shells.
What are the various letters used by Bohr to represent electron shells in an atom?
In the figure given alongside
(a) Name the shells denoted by A,B, and C. Which shell has least energy
(b) Name X and state the charge on it
(c) The above sketch is of …………. Model of an atom

State in brief the drawbacks of Rutherford's atomic model correlating them with the postulates of Bohr’s atomic model.
State true or false. If false, correct the statement.
Smaller the size of the orbit, lower is the energy of the orbit.
Explain the postulates of Bohr’s atomic model.
Which of the following in Fig. 4.2 do not represent Bohr’s model of an atom correctly?
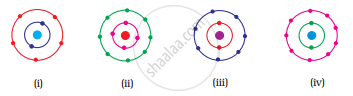
The atomic model based on quantum theory was first proposed by ______.
Bohr's model of atoms ______.
The scientist who proposed the atomic model based on the quantisation of energy for the first time is ______.
