Advertisements
Advertisements
प्रश्न
Which of the following in Fig. 4.2 do not represent Bohr’s model of an atom correctly?
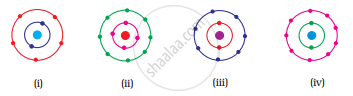
पर्याय
(i) and (ii)
(ii) and (iii)
(ii) and (iv)
(i) and (iv)
Advertisements
उत्तर
(ii) and (iv)
Explanation -
Fig. (ii) contains 4 electrons in K shell and fig. (iv) contains 9 electrons in L shell which are not in accordance with Bohr’s model.
APPEARS IN
संबंधित प्रश्न
Draw a sketch of Bohr’s model of an atom with three shells.
Name the central part of an atom where protons and neutrons are held together.
In the figure given alongside
(a) Name the shells denoted by A,B, and C. Which shell has least energy
(b) Name X and state the charge on it
(c) The above sketch is of …………. Model of an atom

Give the postulates of Bohr's atomic model
State true or false. If false, correct the statement.
Smaller the size of the orbit, lower is the energy of the orbit.
Explain the postulates of Bohr’s atomic model.
An atom with 3 protons and 4 neutrons will have a valency of
What are the Limitations of Bohr’s Model?
The scientist who proposed the atomic model based on the quantisation of energy for the first time is ______.
Given below are two statements:
Statement I: Atoms are electrically neutral as they contain equal number of positive and negative charges.
Statement II: Atoms of each element are stable and emit their characteristic spectrum.
In the light of the above statements, choose the most appropriate answer from the options given below:
