Advertisements
Advertisements
प्रश्न
Which of the following in Fig. 4.2 do not represent Bohr’s model of an atom correctly?
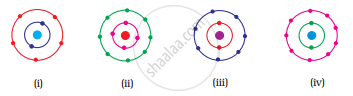
विकल्प
(i) and (ii)
(ii) and (iii)
(ii) and (iv)
(i) and (iv)
Advertisements
उत्तर
(ii) and (iv)
Explanation -
Fig. (ii) contains 4 electrons in K shell and fig. (iv) contains 9 electrons in L shell which are not in accordance with Bohr’s model.
APPEARS IN
संबंधित प्रश्न
Draw a sketch of Bohr’s model of an atom with three shells.
Describe Bohr’s model of the atom.
Name the scientists who described the arrangement of electrons in an atom.
What are the various letters used by Bohr to represent electron shells in an atom?
Describe Bohr's model of the atom. How did Neils Bohr explain the stability of atom?
In the figure given alongside
(a) Name the shells denoted by A,B, and C. Which shell has least energy
(b) Name X and state the charge on it
(c) The above sketch is of …………. Model of an atom

Give the postulates of Bohr's atomic model
Explain the postulates of Bohr’s atomic model.
Niels Bohr was Born on ______.
The scientist who proposed the atomic model based on the quantisation of energy for the first time is ______.
