Advertisements
Advertisements
Question
Give reason for the higher boiling point of ethanol in comparison to methoxymethane.
Advertisements
Solution 1
Ethanol undergoes intermolecular H-bonding due to the presence of the −OH group, resulting in the association of molecules. Extra energy is required to break these hydrogen bonds. On the other hand, methoxymethane does not undergo H-bonding. Therefore, ethanol has a higher boiling point than methoxymethane.
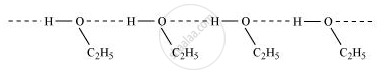
Solution 2
The higher boiling point of ethanol may be attributed to intermolecular hydrogen bonding. The significant bonding in ethanol requires additional energy to dissociate these bonds and transition it into the vapour phase.
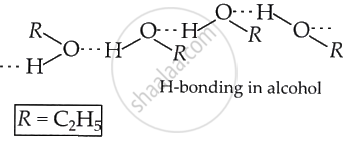
Methoxymethane, an ether, lacks such hydrogen bonding and is therefore a low-boiling liquid.
\[\ce{\underset{between molecules)}{\underset{(lacks H-bonding}{\underset{Methoxymethane}{CH3 - O - CH3}}}}\]
