Advertisements
Advertisements
Question
Give one example of a decomposition reaction which is carried out with electricity.
Advertisements
Solution
Example of the decomposition reaction are as follows:
with electricity:
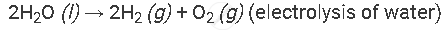
APPEARS IN
RELATED QUESTIONS
What does one mean by exothermic reaction? Give example.
Taking into consideration the relationship in the first pair, complete the second pair
2H2 + O2 → 2H2O :Combination Reaction :: 2HgO → 2Hg + O2:_________
The white solid compound A decomposes quite rapidly on heating in the presence of a black substance X to form a solid compound B and a gas C. When an aqueous solution of compound B is reacted with silver nitrate solution, then a white precipitate of silver chloride is obtained along with potassium nitrate solution. Gas C does not burn itself but helps burn other things.
(a) What is compound A?
(b) What is compound B?
(c) What is gas C?
(d) What do you think is the black substance X? What is its function?
(e) What is the general name of substances like X?
State an important use of decomposition reactions.
Give one example of a decomposition reaction which is carried out by applying heat.
State the change in colour observed in following case mentioning the reason:
Silver chloride is exposed to sunlight.
A metal salt MX when exposed to light splits up to form metal M and a gas X2. Metal M is used in making ornaments whereas gas X2 is used in making bleaching powder. The salt MX is itself used in black and white photography.
(a) What do you think metal M is?
(b) What could be gas X2?
(c) Name the metal salt MX.
(d) Name any two salt solutions which on mixing together can produce a precipitate of salt MX.
(e) What type of chemical reaction takes place when salt MX is exposed to light? Write the equation of the reaction?
Classify the following reaction as combination, decomposition, displacement, precipitation and neutralization. Also balance the equation.
\[\ce{Zn_{(s)} + H2SO4 -> ZnSO4_{(s)} + H2_{(g)}}\]
Explain the following type of chemical reaction, giving two examples for it:
Decomposition reaction
How will you obtain Nitrogen dioxide from lead nitrate.
Also give balanced equations for the reactions
Give a balanced equation for the following type of reaction:
A thermal decomposition reaction in which a metallic nitrate decomposes to give – a basic oxide.
The chemical reaction in which two or more products are formed from a single reactant is called ______ reaction.
What is electrolysis?
Electrolysis is type of ______ reaction.
Chemical volcano is an example of ______ type of reaction.
The following reaction is used for the preparation of oxygen gas in the laboratory:
\[\ce{2KClO3_{(s)} ->[Heat] 2KCl + 3O2_{(g)}}\]
Which of the following statement about the reaction is correct?
Marble statues are corroded or stained rain water. Identify the main reason.

Balance the following chemical equation and identify the type of chemical reaction.
`"HgO"("s") overset("(heat)")(->) "Hg"("l") + "O"_2("g")`
A metal nitrate 'A' on heating gives a metal oxide along with evolution of a brown coloured gas 'B' and a colourless gas, which helps in burning. Aqueous solution of 'A' when reacted with potassium iodide forms a yellow precipitate.
- Identify 'A' and 'B'
- Name the types of the reactions involved in the above statement.
