Advertisements
Advertisements
प्रश्न
Give one example of a decomposition reaction which is carried out with electricity.
Advertisements
उत्तर
Example of the decomposition reaction are as follows:
with electricity:
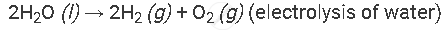
APPEARS IN
संबंधित प्रश्न
What does one mean by exothermic reaction? Give example.
Write one equation for decomposition reactions where energy is supplied in the form of heat.
Taking into consideration the relationship in the first pair, complete the second pair
2H2 + O2 → 2H2O :Combination Reaction :: 2HgO → 2Hg + O2:_________
A solid substance P which is very hard is used in the construction of many buildings, especially flooring. When substance P is heated strongly, it decomposes to form another solid Q and a gas R is given out. Solid Q reacts with water with the release of a lot of heat to form a substance S. When gas R is passed into a clear solution of substance S, then a white precipitate of substance T is formed. The substance T has the same chemical composition as starting substance P.
(a) What is substance P? Write its common name as well as chemical formula.
(b) What is substance Q?
(c) What is gas R?
(d) What is substance S? What is its clear solution known as?
(e) What is substance T? Name any two natural forms in which substance T occurs in nature.
What type of reaction is represented by the following equation?
\[\ce{CaCO3 -> CaCO + CO2}\]
Which of the following can be decomposed by the action of light?
(a) NaCl
(b) KCl
(c) AgCl
(d) CuCl
How can the rate of the chemical reaction, namely, decomposition of hydrogen peroxide be increased?
Answer the following question.
2 g of silver chloride is taken in a china dish and the china dish is placed in sunlight for some time. What will be your observation in this case? Write the chemical reaction involved in the form of a balanced chemical equation. Identify the type of chemical reaction.
Classify the following reaction into –
- Direct combination
- Decomposition
- Displacement
- Double decomposition
The reaction is – Zinc hydroxide on heating gives zinc oxide and water.
Give a balanced equation for –
A double decomposition neutralization reaction involving an acid and a base
Give a balanced equation for the following type of reaction:
A thermal decomposition reaction in which a metallic nitrate decomposes to give – a basic oxide.
Photolysis is a decomposition reaction caused by ______
The following reaction is used for the preparation of oxygen gas in the laboratory:
\[\ce{2KClO3_{(s)} ->[Heat] 2KCl + 3O2_{(g)}}\]
Which of the following statement about the reaction is correct?
On heating blue coloured powder of copper (II) nitrate, in a boiling tube, copper oxide (black), oxygen gas and a brown gas X is formed
- Write a balanced chemical equation of the reaction.
- Identify the brown gas X evolved.
- Identify the type of reaction.
- What could be the pH range of aqueous solution of the gas X?
These consist of two statements – Assertion (A) and Reason (R). Answer these questions selecting the appropriate option given below:
Assertion: Silver bromide decomposition is used in black and white photography.
Reason: Light provides energy for this exothermic reaction.
What is observed when silver chloride is exposed to sunlight? Give the type of reaction involved.
Write the molecular formula of calcium carbonate.
