Advertisements
Advertisements
Question
Give a balanced equation when dilute hydrochloric acid is added to : Zinc Metal
Advertisements
Solution
Zn + 2HCI → ZnCl2 +H2
APPEARS IN
RELATED QUESTIONS
Hydrochloric acid contains (i) hydrogen (ii) chlorine. Prove it. Write equations for the reactions.
Give reason for the following:
silver nitrate solution can be used to distinguish HCl from HNO3
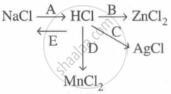
Study the flow chart and give balanced equations with conditions for the conversions A, B, C, D and E.
Give a balanced equation when dilute hydrochloric acid is added to : Calcium carbonate
How will you prove that hydrochloric acid contains
- hydrogen
- chlorine?
Write equations for the reactions.
- Give only one suitable chemical test to identify the following gases.
- Ammonia
- Sulphur dioxide
- Hydrogen chloride
- Chlorine
- Carbon dioxide
- Oxygen
- Hydrogen
- Select a basic gas mentioned in Q. (a). How is the basic nature suspected?
- Select acidic gases from the gases mentioned in Q. (a). How is the acidic nature suspected?
- The two gases A and B are bleaching agents. A is greenish-yellow and bleaches due to its oxidizing property while B is a colourless gas that bleaches due to reduction. Identify A and B?
- Which gas turn blue cobalt chloride paper light pink? Give one similarity in the test between
- Cl2 and HCl
- SO2 and CO2.
Complete and balance the following reaction, state whether dilute or conc. acid is used.
\[\ce{NH4OH + HCl->}\]
Complete and balance the following reaction, state whether dilutes or conc. acid is used.
\[\ce{NH4OH + HCl -> }\]
Complete and balance the following reaction, state whether dilute or conc. acid is used.
NH4OH + HCl ⟶
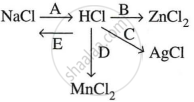
Study the flow chart and give balanced equations with conditions for the conversions A, B, C, D and E.