Advertisements
Advertisements
Question
Figure shows the stopping potential (V0) for the photo electron versus (1/λ) graph, for two metals A and B, λ being the wavelength of incident light.
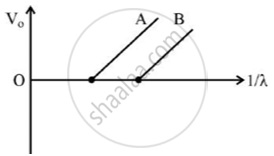
- How is the value of Planck’s constant determined from the graph?
- If the distance between the light source and the surface of metal A is increased, how will the stopping potential for the electrons emitted from it be effected? Justify your answer.
Justify
Long Answer
Advertisements
Solution
a. Planck’s constant can be determined from the gradient of the graph.
Gradient = `(h c)/e`
h = Planck’s constant
c = Velocity of light in vacuum
e = Charge of electron
The gradient is to be measured from the graph. The values of c and e are known. Hence, h can be determined.
b. As the distance between the light source and the surface of metal A increases, the intensity of the incident light diminishes. However, the stopping potential is independent of the intensity of the incident light. The stopping potential will remain unaffected.
shaalaa.com
Is there an error in this question or solution?
