Advertisements
Advertisements
Question
Explain why:
Fused CaCl2 or conc. H2SO4 is used in a desiccator.
Advertisements
Solution
Fused CaCl2 or concentrated H2SO4 is deliquescent in nature, i.e. it absorbs moisture, and hence, these are used in desiccators as drying agents.
APPEARS IN
RELATED QUESTIONS
Give balanced chemical equation for the reaction of water with Sodium oxide.
State whether the given statement are true or false. If true, write the reason.
Ganga−Brahmaputra plain of India is an overpopulated region.
State whether the given statement are true or false. If true, write the reason.
Water availability per person in India is declining.
How is aquatic life benefited by the fact that water has maximum density at 4oC?
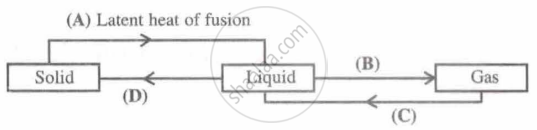
Identify A, B, C and D; first one is done for you.
Explain why oil spills have an adverse effect on marine life.
True or false? Correct the wrong statement.
We get rainwater throughout the year.
Pour water from the ice tray into a kettle. Ask an elder at your home to heat the kettle and observe the mouth of the kettle when the water starts boiling.
What do you see?
A jumbled word is given here. Write the correct form of the word.
CHARREGE
Why should we not throw used tea leaves into sink?
