Advertisements
Advertisements
Question
Explain in brief the experimental proof which led to the discovery of –
Electrons
Advertisements
Solution
Electrons: English physicist William Crookes in 1878 discovered the cathode rays
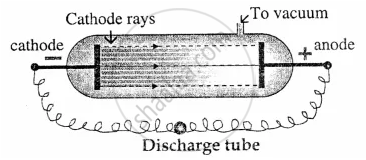
He found that when an electric discharge is passed through a tube containing a gas at low pressure (0.01 mm of Hg).
Blue rays were emitted from the negative plate [i.e. cathode]. These rays were called cathode rays and consist of negatively charged particles now called electrons. Electrons are an essential part of each and every atom.
APPEARS IN
RELATED QUESTIONS
The ion of an element has 3 positive charges. The mass number of atom of this element is 27 and the number of neutrons is 14. What is the number of electrons in the ion?
What are cathode rays? How are these rays formed?
Which sub-atomic particle was discovered by Chadwick.
If electrons are getting added to en element Y; then
Is Y getting oxidized or reduced?
Name or state the following:
Ion formed by the gain of an electron(s)
In the formation of (i) oxygen molecule (ii) carbon tetrachloride molecule, state the following:
- Electronic configuration of nearest inert gas attained.
- How many electrons are shared/transferred in bond formation
- Which type of bonds these compounds form?
- Draw their orbital diagrams.
What are the noble gases?
_______ is a negatively charged particle.
Anode rays travel in straight lines.
Helium has two electrons in the outermost orbit and so it is chemically inert.
