Advertisements
Advertisements
प्रश्न
Explain in brief the experimental proof which led to the discovery of –
Electrons
Advertisements
उत्तर
Electrons: English physicist William Crookes in 1878 discovered the cathode rays
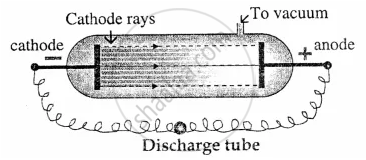
He found that when an electric discharge is passed through a tube containing a gas at low pressure (0.01 mm of Hg).
Blue rays were emitted from the negative plate [i.e. cathode]. These rays were called cathode rays and consist of negatively charged particles now called electrons. Electrons are an essential part of each and every atom.
APPEARS IN
संबंधित प्रश्न
Fill in the blank of the following statement :
Cathode rays are a beam of fast moving _________.
An electron carries a ________ charge.
Which sub-atomic particle was discovered by Chadwick.
Write down the number of electrons in X and neutrons in Y.
An atom X has three electrons more than the noble gas configuration. What type of ion will it form?
If electrons are getting added to en element Y; then
What charge will Y migrate to during the process of electrolysis?
Choose the correct answer from A, B, C and D:
When a metal atom becomes an ion:
What is the term defined below?
A bond formed by a shared pair of electrons, each bonding atom contributing one electron to the pair.
On the basis of Thomson’s model of an atom, explain how the atom is neutral as a whole.
Rutherford’s ‘alpha (α) particles scattering experiment’ resulted in to discovery of
