Advertisements
Advertisements
Question
Explain Bredig’s arc method.
Advertisements
Solution
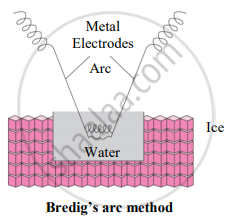
- Colloidal sols can be prepared by electrical disintegration using Bredig’s arc method.
- This process involves vaporization as well as condensation.
- Colloidal sols of metals such as gold, silver, platinum can be prepared by this method.
- In this method, an electric arc is struck between electrodes of metal immersed in the dispersion medium.
- The intense heat produced vapourizes the metal which then condenses to form particles of colloidal sol.
APPEARS IN
RELATED QUESTIONS
Distinguish between lyophobic and lyophilic sols.
Write a note on the Tyndall effect.
Write a note on Brownian motion.
Write a note on Types of emulsion.
Explain micelle formation in a soap solution.
Draw labelled diagram Tyndall effect.
Draw labelled diagram Dialysis.
Draw labelled diagram of Soap micelle.
Which one of the following methods is used to make platinum sol?
Mist is a colloidal solution of ____________.
Which of the following statements is CORRECT?
Which of the following is an example of liquid-liquid system?
The precipitation power of an electrolyte increases with ______.
Which of the following is NOT true for lyophilic colloids?
Mixing of two oppositely charged sols leads to ____________.
Which of the following is multimqlecular colloid?
Which of the following ionic species has highest precipitating power?
Freshly prepared precipitate sometimes gets converted to colloidal solution by ______.
Which of the following electrolytes will have maximum coagulating value for AgI/Ag+ solution?
Which of the following process is responsible for the formation of delta at a place where rivers meet the sea?
Match the items of Column I and Column II.
| Column I | Column II |
| (i) Butter | (a) dispersion of liquid in liquid |
| (ii) Pumice stone | (b) dispersion of solid in liquid |
| (iii) Milk | (c) dispersion of gas in solid |
| (iv) Paints | (d) dispersion of liquid in solid |
Which of the following substances will precipitate the negatively charge emulsion
The size of a raw mango shrinks to a much smaller size when kept in a concentrated salt solution. Which one of the following processes can explain this?
The migration of dispersion medium under the influence of an electric potential is called ______.
In which of the following, the Tyndall effect is not observed?
Which of the following electrolytes will be most effective in coagulating negative sol?
Identify positively charged sol from following.
