Advertisements
Advertisements
Question
Draw the structure of major monohalo products in the following reaction:

Advertisements
Solution
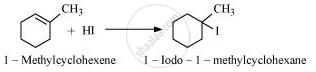
APPEARS IN
RELATED QUESTIONS
Draw the structures of major monohalo products in each of the following reaction:

Draw the structure of major monohalo products in the following reaction:

Name the following halide according to the IUPAC system and classify it as an alkyl, allyl, benzoyl (primary, secondary, tertiary), vinyl or aryl halide:
CH3C(Cl)(C2H5)CH2CH3
Name the following halide according to the IUPAC system and classify it as an alkyl, allyl, benzoyl (primary, secondary, tertiary), vinyl or aryl halide:
m-ClCH2C6H4CH2C(CH3)3
Name the following halide according to the IUPAC system and classify it as an alkyl, allyl, benzoyl (primary, secondary, tertiary), vinyl or aryl halide:
o-Br-C6H4CH(CH3)CH2CH3
The position of \[\ce{-Br}\] in the compound in \[\ce{CH3CH = CH(Br)(CH3)2}\] can be classified as ______.
What should be the correct IUPAC name for diethylbromomethane?
Which of the carbon atoms present in the molecule given below are asymmetric?
\[\begin{array}{cc}
\phantom{}\ce{HO}\phantom{.....}\ce{OH}\phantom{..}\ce{H}\phantom{.....}\ce{O}\phantom{..}\\
\phantom{..}\backslash\phantom{.....}|\phantom{....}|\phantom{.....}//\phantom{.}\\
\ce{\overset{a}{C} - \overset{b}{C} - \overset{c}{C} - \overset{d}{C}}\\
\phantom{..}//\phantom{.....}|\phantom{....}|\phantom{....}\phantom{.}\backslash\phantom{...}\\
\phantom{}\ce{O}\phantom{......}\ce{H}\phantom{...}\ce{OH}\phantom{...}\ce{H}\phantom{}\\
\end{array}\]
Two isomers (A) and (B) with molar mass 184 g/mol and elemental composition C 52.2%; H 4.9% and Br 42.9% gave benzoic acid and p-bromobenzoic acid, respectively on oxidation with KMnO4. Isomer ‘A’ is optically active and gives a pale yellow precipitate when warmed with alcoholic AgNO3. Isomer ‘A’ and ‘B’ are, respectively:
Name the following halide according to IUPAC system and classify it as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halide:
\[\ce{CH2CH2C(CH3)2CH2I}\]
Name the following halide according to the IUPAC system and classify them as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl, or aryl halide:
\[\ce{CH3CH2C(CH3)2CH2I}\]
Name the following halide according to IUPAC system and classify them as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halide:
CH3C(Cl)(C2H5)CH2CH3
Write the structure of the following organic halogen compound.
1,4-Dibromobut-2-ene
Name the following halide according to IUPAC system and classify them as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halides:
\[\ce{CH3C(Cl)(C2H5)CH2CH3}\]
Name the following halide according to IUPAC system and classify them as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halides:
\[\ce{CH3CH2C(CH3)2CH2I}\]
Name the following halide according to IUPAC system and classify them as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halides:
\[\ce{CH3 CH2 C(CH3)2 CH2I}\]
Name the following halides according to IUPAC system and classify them as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halides:
\[\ce{CH3C(C2H5)2CH2Br}\]
Draw the structure of major monohalo products in the following reaction:

IUPAC name of 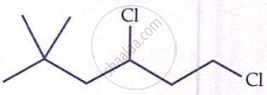 is ______.
is ______.
