Advertisements
Advertisements
Question
Describe an experiment to prove the following:
HCI is highly soluble in water.
Advertisements
Solution
The high solubility of HCI gas can be demonstrated by "Fountain experiment''. In it, a well dried round bottom flask is filled with dry HCI gas and fitted with a two holed rubber stopper. A jet tube is inserted into the flask through one of the holes. A dropper filled with water is fitted into the other hole. Closing the outer end of the jet tube, the flask is inverted and the tube is dipped into a blue litmus solution taken in a beaker, then dropper is pressed. It is seen that the blue litmus solution enters the jet tube with a great force, forming a red fountain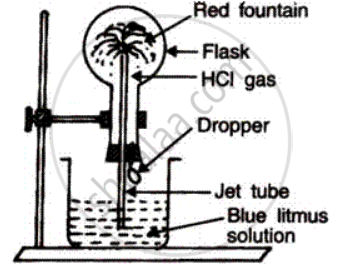
It happens because water introduced from dropper completely absorbs HCI gas, thereby creating a very low pressure within the flask. To make up for this loss in pressure, blue litmus solution rises in the jet tube and colour of litmus changes to red.
APPEARS IN
RELATED QUESTIONS
How will you prepare hydrochloric acid in laboratory? OR
Describe briefly the method employed to dissolve hydrogen chloride gas in water as it is prepared. What is the purpose of funnel while preparing hydrochloric acid from HCI gas?
Explain, why (or give reasons for)
Hydrochloric acid cannot be concentrated above 22.2% by boiling.
Write the uses of hydrochIoric acid.
How can you prove that hydrochloric acid contain :
Hydrogen
Write equation for the reaction between hydrochloric acid of the following: Manganese dioxide
What wilI you observed when concentrated HCI is added to lead (IV) oxide and warmed ?
Aqua regia is a mixture of ______.
In the laboratory preparation of hydrochloric acid, HCl gas is dissolved in water.
- Draw a diagram to show the arrangement used for the absorption of HCl gas in water.
- State why such an arrangement is necessary? Give two reasons for the same.
- Write the chemical equation for the laboratory preparation of HCl gas when the reaction is:
- Below 200°C
- Above 200°C
State one appropriate observation of the following:
Which gas is evolved when potassium sulphite with dilute hydrochloric acid.
Name the anion present in the following compound:
The action of heat on the insoluble compound C produces a gas which turns lime water turbid.
