Advertisements
Advertisements
Question
Describe an experiment to demonstrate that air exerts pressure.
Advertisements
Solution
Experiment to demonstrate that air exerts pressure:
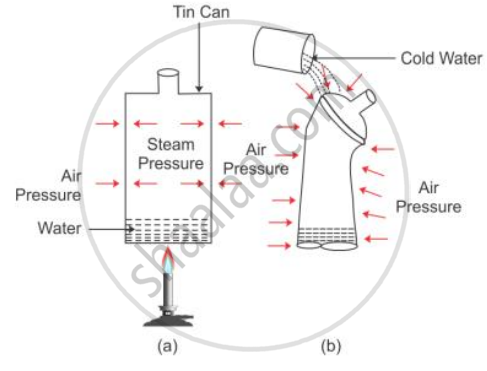
Take a thin can fitted with an airtight stopper. The stopper is removed and a small quantity of water is boiled in the can. Gradually the steam occupies the entire space of can by expelling the air from it [Fig (a)]. Then stopper is then tightly replaced and simultaneously the flame beneath the can is removed. Cold water is then poured over the can.
It is observed that the can collapses inwards as shown in fig (b).
The reason is that the pressure due to steam inside the can is same as the air pressure outside the can [Fig (a)]. However, on pouring cold water over the can, fitted with a stopper [fig (b)], the steam inside the can condenses producing water and water vapour at very low pressure. Thus, the air pressure outside the can becomes more than the vapour pressure inside the closed can.
Consequently, the excess atmospheric pressure outside the can causes it to collapse inwards.
APPEARS IN
RELATED QUESTIONS
When we press the bulb of a dropper with its nozzle kept in water, air in the dropper is seen to escape in the form of bubbles. Once we release the pressure on the bulb, water gets filled in the dropper. The rise of water in the dropper is due to
What do you mean by atmospheric pressure?
How does the atmospheric pressure change with altitude?
We do not feel uneasy even under enormous pressure of the atmosphere above as well as around us. Give a reason.
What is an aneroid barometer? Draw a neat and labelled diagram to explain its construction and working.
How is the reading of a barometer affected when it is taken to (i) a mine, and (ii) a hill?
The atmospheric pressure at earth's surface is P1 and inside mine is P2. They are related as :
Briefly explain the three chief types of winds.
A solid weighs 2.10 N in air. It has a relative density of 8.4. How much will the body weigh if placed: In water
Why do Scuba divers wear special suits and equipment?
