Advertisements
Advertisements
प्रश्न
Describe an experiment to demonstrate that air exerts pressure.
Advertisements
उत्तर
Experiment to demonstrate that air exerts pressure:
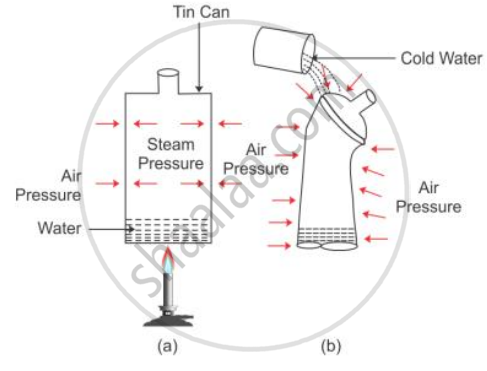
Take a thin can fitted with an airtight stopper. The stopper is removed and a small quantity of water is boiled in the can. Gradually the steam occupies the entire space of can by expelling the air from it [Fig (a)]. Then stopper is then tightly replaced and simultaneously the flame beneath the can is removed. Cold water is then poured over the can.
It is observed that the can collapses inwards as shown in fig (b).
The reason is that the pressure due to steam inside the can is same as the air pressure outside the can [Fig (a)]. However, on pouring cold water over the can, fitted with a stopper [fig (b)], the steam inside the can condenses producing water and water vapour at very low pressure. Thus, the air pressure outside the can becomes more than the vapour pressure inside the closed can.
Consequently, the excess atmospheric pressure outside the can causes it to collapse inwards.
APPEARS IN
संबंधित प्रश्न
The atmospheric pressure is nearly 105 Pa.
Write the numerical value of the atmospheric pressure on the earth surface in pascal.
Write the numerical value of the atmospheric pressure on the surface of earth in pascal.
What is a barometer?
State two uses of a barometer.
Convert 1 mm of Hg into pascal. Take density of Hg = 13.6 x 103 kg m-3 and g = 9.8 m s-2 .
The highest air pressure at sea level was recorded at ______ Russia.
The small variations in pressure that exist largely determine the ______ and ______ patterns of the earth.
Tyre pressure of 30psi is almost ______ the atmospheric pressure.
Why do Scuba divers wear special suits and equipment?
