Advertisements
Advertisements
Question
Derive the expression for the work done in a volume change in a thermodynamic system.
Advertisements
Solution
Work done in volume changes: Consider a gas contained in the cylinder fitted with a movable piston. Suppose the gas is expanded quasi-statically by pushing the piston by a small distance dx. Since the expansion occurs quasi-statically the pressure, temperature and internal energy will have unique values at every instant.
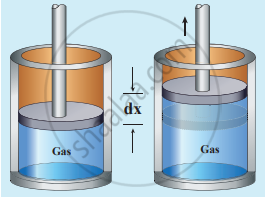
Work done by the gas
The small work done by the gas on the piston
dW = Fdx ….........(1)
The force exerted by the gas on the piston F = PA.
Here A is the area of the piston and P is the pressure exerted by the gas on the piston.
Equation (1) can be rewritten as Work done by the gas
dW = PA dx …..........(2)
But Adx = dV= change in volume during this expansion process.
So the small work done by the gas during the expansion is given by
dW = PdV .........….(3)
dV is positive since the volume is increased. Here, dW is positive.
In general, the work done by the gas by increasing the volume from Vi to Vf is given by
W = `int_("V"_"i")^("V"_"f") "PdV"` ..........(4)
Suppose if the work is done on the system, then Vi > Vf. Then, W is negative.
Note here the pressure P is inside the integral in equation (4). It implies that while the system is doing work, the pressure need not be constant. To evaluate the integration we need to first express the pressure as a function of volume and temperature using the equation of state.
APPEARS IN
RELATED QUESTIONS
The work done in vacuum when 300 m mole of an ideal gas expands until its volume is increased by 2.3 dm3 at 1 bar pressure is ______ mole.
An ideal gas is taken from (Pi, Vi) to (Pf, Vf) in three different ways. Identify the process in which the work done on the gas the most.

Define the internal energy of the system.
Give the sign convention for Q and W.
Give the expression for work done by the gas.
Explain Joule’s Experiment of the mechanical equivalent of heat.
An ideal gas is allowed to expand in a well insulated container against a constant external pressure of 2.5 bar from an initial volume of 2.50 dm3 to a final volume of 4.50 dm3. The change in internal energy (∆U) of the gas will be ______.
An ideal gas expands against a constant external pressure of 2.0 atmosphere from 20 litre to 40 litre and absorbs 10 kJ of heat from the surrounding. What is the change in the internal energy of the system?
(Given: 1 atm-litre = 101.3 J)
The value of Δ U is ______ when 2 kJ of heat is released and 6 kJ of work is done on the system.
Calculate the change in internal energy of a gas if a gas is compressed adiabatically from 25 dm³ to 15 dm³ at constant external pressure of 4 bar.
